全文HTML
--> --> -->CsPbIBr2薄膜的制备方法有多种, 包括一步旋涂法、两步旋涂法以及气相沉积法等[20-23]. 其中一步旋涂法由于操作简单、成本低是目前最常用的制备方法, 但是CsPbIBr2薄膜质量通常较差, 其电池效率仍有很大的提升空间[24,25]. 主要原因之一在于传统的一步旋涂法下CsPbIBr2前驱体液通常选择卤素溶解度较好的纯DMSO溶剂, 溶质以CsI-DMSO-PbBr2的中间体形式存在[26,27]. 在薄膜制备过程中, 大量的中间体阻碍了CsI与PbBr2的反应, 且DMSO挥发温度较高, 直接影响了CsPbIBr2的结晶速度和薄膜质量, 还会促进离子的迁移过程, 导致非辐射复合现象发生[28,29]. 因此, 在DMSO溶剂体系下如何控制DMSO残余量, 从而促进钙钛矿结晶过程是提高薄膜质量的关键. Zhang等[30]在旋涂后对薄膜进行光照处理, 为DMSO分子的扩散提供了额外的能量, 从而促进了CsPbIBr2晶粒的生长, 实验发现AM1.5 G下持续照射60 min时钙钛矿薄膜结晶质量最好, 晶粒平均尺寸达到380 nm; Zhu等[31]在低温退火条件下研究了DMSO作为溶剂时前驱体溶液不同熟化时间下CsPbIBr2的结晶过程, 发现随着熟化时间的延长钙钛矿前驱体薄膜逐渐由透明向黄色转变, 且退火温度可以降低至100 °C, 熟化时间延长至3周时, 制备的钙钛矿电池在效率达到6.55%. 调整钙钛矿前驱体溶液旋涂过程中DMSO的残留量也可以促进晶粒增长. Zhang等[32]对比了6种反溶剂对于CsPbIBr2结晶质量的影响, 并引入有机阳离子修饰CsPbIBr2与Spiro-OMeTAD间界面. 实验结果发现二乙醚作为反溶剂时钙钛矿薄膜结晶度最好, 以Au作为电极制备的CsPbIBr2电池效率达到了9.17%; Bian等[33]以IPA作为反溶剂, 并在异丙醇中加入不同浓度的甲醚碘(FAI)来提高钙钛矿前驱体溶液中的反应速率, 得到的CsPbIBr2薄膜孔洞较少且晶粒尺寸提高到300—600 nm.
受以上观点启发, 考虑到常规反溶剂法工艺窗口较窄、对于滴加量和滴加方式都需要精确控制, 不适用于规模化商业生产, 为此我们提出一种前驱体膜处理工艺以制备高效稳定的碳基CsPbIBr2电池. 以异丙醇(IPA)作为反溶剂, 通过调控前驱体膜中DMSO的残留量使其快速达到过饱和状态, 从而促进钙钛矿成核及生长. 此外, 为了进一步提高薄膜质量, 我们在IPA中加入了硫氰酸胍(C2H4N4S)作为钝化剂并调控钙钛矿的结晶行为. C2H4N4S中的氨基(—NH2)与钙钛矿中的卤素离子形成了氢键作用能够有效抑制离子的迁移, 从而改善了结晶过程. SCN–是一种类卤化物离子, 与钙钛矿中的Pb2+存在较强配位作用, 有助于溶液中溶质的聚集, 从而降低成核密度, 而且前驱体膜中引入SCN–能够促进中间体相的分解, 进而改善了钙钛矿的结晶过程[34,35]. 此外, 与界面修饰只能钝化钙钛矿薄膜表面缺陷相比, 在前驱体膜中引入钝化剂能够有效减少薄膜不同深度下的晶界和缺陷. 实验结果表明, 前驱体膜处理工艺下得到的CsPbIBr2薄膜晶粒尺寸有明显提高, 平均晶粒尺寸达到800 nm, 制备的碳基电池效率从未处理的5.29%提高至6.71%, 提高了近21.16%. 本研究旨在研究纯DMSO溶剂体系下全无机钙钛矿前驱体膜处理过程中反溶剂与钝化剂的协同作用, 为改进碳基CsPbIBr2器件性能提供理论参考.
2.1.材 料
碘化铯(CsI, 99.98%)、溴化铅(PbBr2, 99.99%)、二甲基亚砜(DMSO, 分析纯)、硫氰酸胍(C2H4N4S, 99.5%), 以上药品采购于西安宝莱特光电科技有限公司; SnO2胶体溶液(15 wt%水溶液), 以上药品采购于阿法埃莎公司; 异丙醇(IPA, 分析纯), 以上药品采购于上海阿拉丁生化科技股份有限公司; 导电碳浆采购于上海迈拓崴化工新材料科技有限公司.2
2.2.器件制备
FTO导电玻璃(尺寸为2 cm × 2 cm)依次通过去离子水、丙酮、异丙醇、乙醇超声清洗20 min, 然后将玻璃片进行等离子体处理15 min以进一步改善表面亲水性.SnO2胶体溶液用去离子水稀释到2.67%的体积浓度, 以2000 r/min旋转30 s沉积到FTO基底上, 然后在150 ℃温度下退火30 min; 完成后用等离子体处理15 min.
在手套箱中配制CsPbIBr2钙钛矿前驱体溶液, 将440 mg PbBr2 和312 mg CsI溶解在1 ml DMSO中, 然后在60 ℃下搅拌至完全溶解, 并用0.45 μm的PTFE滤头过滤溶液. 采用传统一步法制备CsPbIBr2薄膜, 将90 μl钙钛矿前驱体液以600 r/min旋涂5 s、3200 r/min旋涂45 s, 随后将前驱体薄膜静置2 min并滴加40 μL IPA以及40 μL溶有不同浓度C2H4N4S的IPA溶液, 在相同旋涂参数下再次旋涂, 然后将玻璃片放置260 ℃热台上退火10 min.
最后, 采用刮涂法制备碳电极. 用高温胶带将玻璃片固定后, 使用刮刀将碳浆均匀涂覆在钙钛矿薄膜上, 其厚度通过胶带控制, 面积为0.2 cm2, 然后将器件置于120 ℃热台上退火15 min.
2
2.3.表征测试
样品形貌结构利用扫描电镜(SEM, S-4800)、原子力显微镜(AFM, Dimension 3100 Veeco)进行表征; 样品光学分析采用紫外-可见分光光谱(UV-Vis, PerkinElmer UV WinLab Lambda 35)、光致发光光谱(PL, F-7000 FL Spectrophotometer)、时间分辨荧光光谱(TRPL, Edinburgh Instruments FLS920 fluorescence spectrometer)进行表征; 样品晶型结构以及元素化学态通过X射线衍射仪(XRD, Rigaku D/MAX 2500)和X射线光电子能谱(XPS, 赛默飞EscaLab Xi+)进行表征.器件光电性能在模拟AM 1.5G照射、光照强度100 mW/cm2 条件下测试(Enlitech SS-F7-3A); 器件光电转换效率(EQE)由Enlitech QE-R进行测试; 器件电化学交流阻抗(EIS)在电化学工作站(CHI760E)上测试.
 图 1 (a) CsPbIBr2薄膜制备流程图; (b) 钙钛矿太阳能电池器件结构示意图; (c) 钙钛矿太阳能电池SEM截面图
图 1 (a) CsPbIBr2薄膜制备流程图; (b) 钙钛矿太阳能电池器件结构示意图; (c) 钙钛矿太阳能电池SEM截面图Figure1. (a) Schematic process for the preparation of CsPbIBr2; (b) schematic of architecture; (c) cross-sectional SEM view of the device structure.
三种方法制备的前驱体薄膜退火前的照片如图2所示. 传统制备方法下, 未退火的前驱体薄膜呈无色透明状(图2), 此时薄膜内组分以DMSO、CsI、PbBr2和CsI-DMSO-PbBr2中间体相为主. 经过IPA处理后的前驱体膜变为浅粉色(图2(b)), 其颜色与CsPbIBr2晶体相近, 说明此时已经发生了少部分钙钛矿成核及结晶过程. 而添加C2H4N4S后前驱体膜中的中间体相更容易分解, 提高了CsPbIBr2成核及结晶程度, 因此薄膜颜色呈棕黄色(图2(c)).
 图 2 不同制备工艺下前驱体薄膜表面照片 (a) 传统方法; (b) IPA处理; (c) IPA处理、C2H4N4S钝化(0.4 mg/ml)
图 2 不同制备工艺下前驱体薄膜表面照片 (a) 传统方法; (b) IPA处理; (c) IPA处理、C2H4N4S钝化(0.4 mg/ml)Figure2. Optical images of precursor film under different preparation processes: (a) conventional method; (b) adding IPA solution; (c) adding IPA solution with 0.4 mg/ml of C2H4N4S.
为验证以上观点, 通过SEM表面测试分析三种制备工艺对于CsPbIBr2薄膜形貌的影响. 如图3(a)—3(c)所示, 可以明显看出传统方法制备的CsPbIBr2薄膜由于退火时前驱体膜中的DMSO溶剂残留量大, 形成的CsI-DMSO-PbBr2中间体相阻碍了前驱体溶质之间的成核及结晶过程, 随着DMSO的不均匀蒸发, 钙钛矿薄膜出现了许多孔洞, 晶粒平均尺寸为420 nm. 当前驱体膜经过IPA处理后, 有效调控了DMSO的残留量及蒸发过程, 前驱体薄膜快速进入过饱和状态, 形核位置的更加平均. 这是由于传统方法下前驱体膜蒸发速率较低, 液膜内微观温度场梯度变化迟缓, 造成钙钛矿成核位置优先在薄膜上方界面处, 从而内部容易出现孔洞及晶界. 而经IPA处理后, 前驱体膜蒸发速率明显提升, 液膜内部温度场自下而上变化明显, 液膜表面及内部均成为成核位置; 在加入C2H4N4S修饰后, 晶粒平均尺寸增长显著, 达到800 nm左右, 且薄膜表面晶界有所减少. 但随着C2H4N4S浓度的进一步提高, 前驱体膜中溶质之间反应速率过快导致了非均匀成核, 从而形成了小的孔洞和晶界, 薄膜质量有所下降, 如图3(d)所示.
 图 3 不同制备工艺下CsPbIBr2薄膜表面SEM图 (a) 传统方法; (b) IPA处理; (c) IPA处理、C2H4N4S钝化(0.4 mg/ml); (d) IPA处理、C2H4N4S钝化(0.8 mg/ml); 对应器件截面SEM图 (e?h)
图 3 不同制备工艺下CsPbIBr2薄膜表面SEM图 (a) 传统方法; (b) IPA处理; (c) IPA处理、C2H4N4S钝化(0.4 mg/ml); (d) IPA处理、C2H4N4S钝化(0.8 mg/ml); 对应器件截面SEM图 (e?h)Figure3. SEM images of CsPbIBr2 film under different preparation processes: (a) Conventional method; (b) adding IPA solution; (c) adding IPA solution with 0.4 mg/ml of C2H4N4S; (d) adding IPA solution with 0.8 mg/ml of C2H4N4S; (e?h) corresponding crosssectional SEM images.
如图3(e)—3(h)为电池器件SEM截面图, 不同制备方法下钙钛矿薄膜厚度接近235 nm, 传统一步法下前驱体薄膜通过蒸发来使溶液达到过饱和状态, 溶质倾向于富集在前驱体薄膜表面. 此时大量的DMSO促使最初形成的晶核不断生长, 由于浓度差作用使该晶核局部区域浓度场进一步降低, 且富集更多溶质, 导致其他晶核区域没有足够溶质供晶核继续生长, 从而制备CsPbIBr2薄膜不同深度处均存在不同程度的晶界和缝隙. 这种现象在经IPA处理后得到一定改善, 而在IPA中加入了C2H4N4S后钙钛矿薄膜更加致密. 但随着C2H4N4S浓度的提升, 促进了前驱体薄膜中溶质的团聚, 导致钙钛矿薄膜晶界及孔隙数量增加.
C2H4N4S加入后CsPbIBr2薄膜质量提升的机理可以解释如下: 1)由于SCN–与Pb2+存在的配位作用, 使得前驱体膜中的中间体相及溶质会发生聚集效应, 与纯IPA处理方法相比, 经C2H4N4S修饰后既能保证前驱体膜中成核点位的平均分布又能降低晶核密度. 2) DMSO与Pb2+的配位强度要弱于SCN–, 导致在前驱体薄膜的旋涂过程中CsI-DMSO-PbBr2中间体相更容易分解, 进而促进了钙钛矿形核和晶粒的快速生长. 3) C2H4N4S中的-NH2与CsPbIBr2中的I–和Br–形成的氢键抑制了离子向晶界迁移, 进一步降低了薄膜缺陷.
为研究前驱体膜处理工艺对于钙钛矿晶体结构的影响, 我们对三种方法制备的CsPbIBr2薄膜进行了XRD表征, 结果如图4(a)所示. 可以看到3个样品均具有比较明显的CsPbIBr2特征峰, 衍射角及对应晶面分别是15.02°(100), 21.32°(110), 26.72°(111), 30.32°(200). 虽然第3种方法引入了新的添加剂, 但由于C2H4N4S剂量很少, 因此对整体钙钛矿薄膜的结构不会造成明显影响. 值得注意的是, 经前驱体薄膜工艺处理后, 两个样品在(100)和(200)晶面的衍射峰强度比值有了显著提高, 由传统方法制备的参比样品的0.4提高至0.71和0.83, 说明晶粒在(100)晶面的生长趋势有所增强.
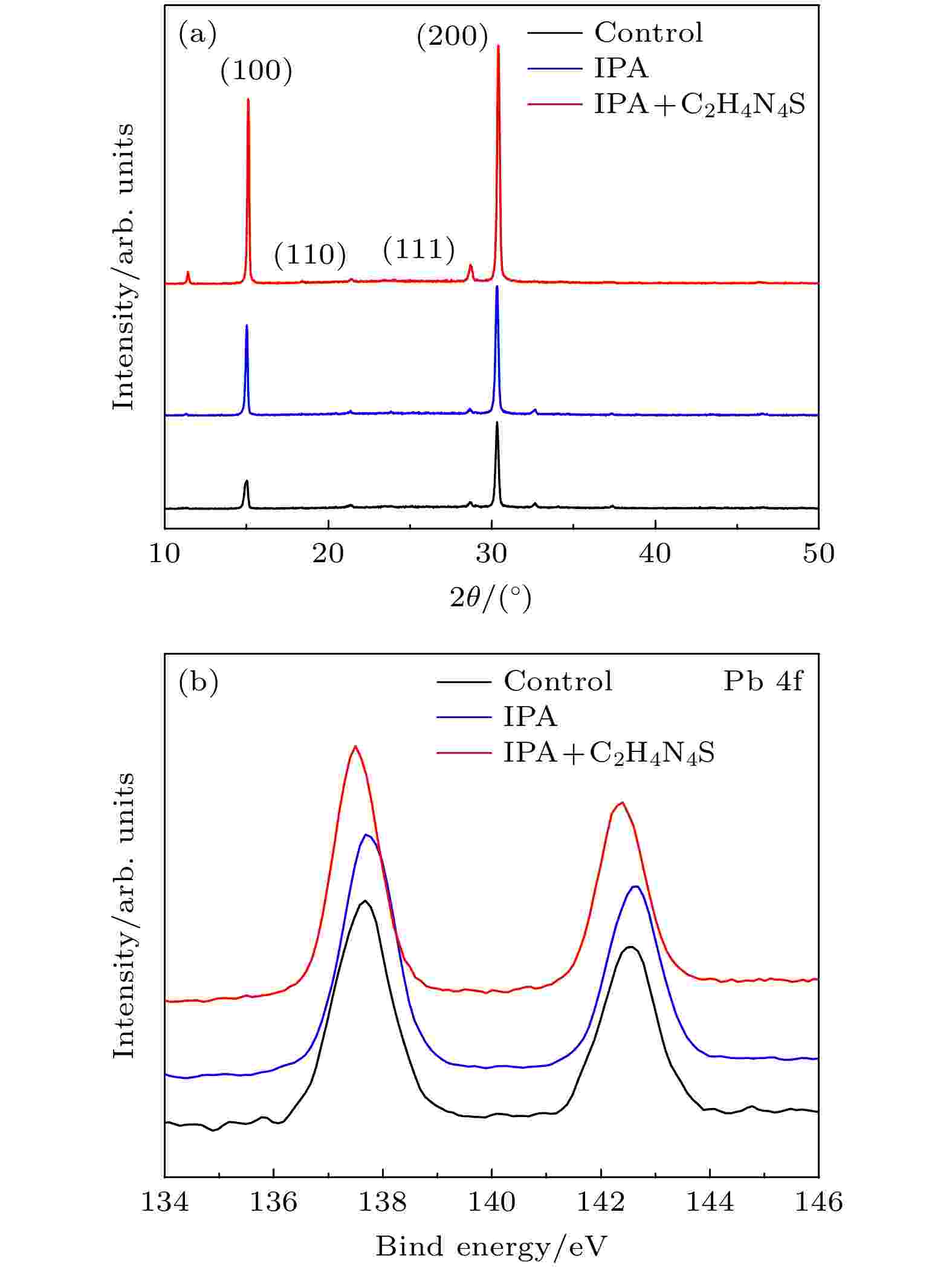 图 4 不同制备工艺下CsPbIBr2薄膜 (a) XRD谱图; (b) XPS谱图
图 4 不同制备工艺下CsPbIBr2薄膜 (a) XRD谱图; (b) XPS谱图Figure4. CsPbIBr2 film under different preparation processes: (a) XRD patterns; (b) XPS spectrum.
为了更深入理解添加剂对于薄膜缺陷的钝化机理, 我们对CsPbIBr2薄膜进行了XPS谱图测试, 如图4(b)所示. 参比样品中Pb 4f7/2和Pb 4f5/2峰值位置分别对应137.7 eV和142.6 eV, IPA反溶后的薄膜峰值位置与参比样品一致, 但C2H4N4S钝化后两个峰的能级下降至137.5 eV和142.4 eV. 能级变化的原因在于Pb原子周围的电子云密度提高, 说明C2H4N4S中的—SH作为路易斯碱与CsPbIBr2中的Pb2+发生了较强的配位作用.
不同制备方法下CsPbIBr2薄膜紫外吸收光谱如图5(a)所示, 3个样品展示了相似的吸收带边(584 nm), 对应禁带宽度为2.12 eV(图5(b)). 对比发现经过前驱体膜处理后的薄膜吸收强度有少许增强, 从而生成更多的载流子, 说明薄膜具有更好的均匀性以及结晶性.
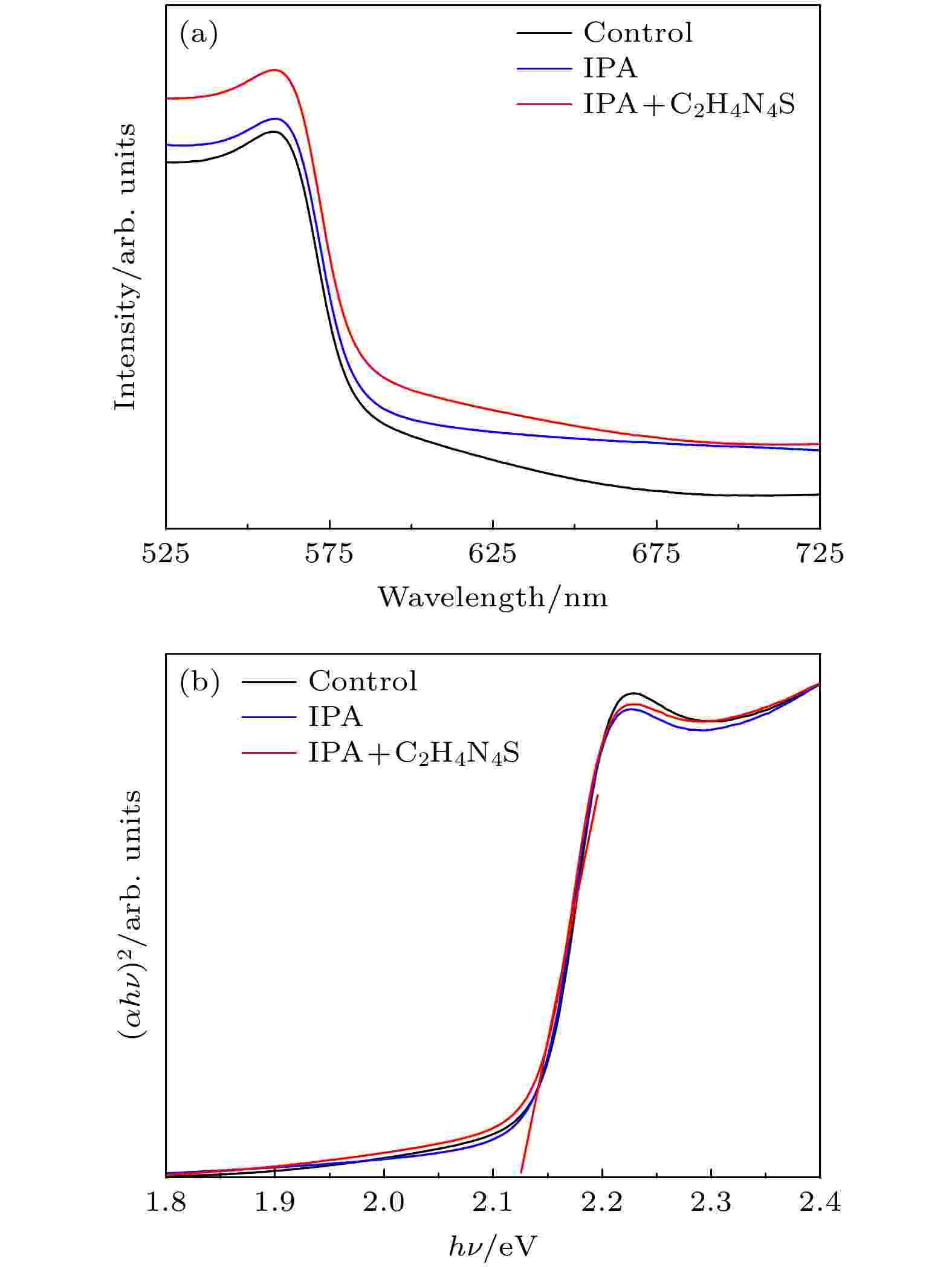 图 5 不同制备工艺下CsPbIBr2薄膜 (a) UV-Vis谱图; (b) Tauc plot谱图
图 5 不同制备工艺下CsPbIBr2薄膜 (a) UV-Vis谱图; (b) Tauc plot谱图Figure5. CsPbIBr2 film under different preparation processes: (a) UV-vis absorbance spectra; (b) Tauc plots.
FTO/CsPbIBr2结构下薄膜的发光光谱如图6(a)所示. 与未处理的钙钛矿薄膜相比, IPA反溶处理后的薄膜发光强度有所提高, 而经过C2H4N4S修饰后的薄膜发光强度最高. 说明前驱体膜处理方法能够有效降低薄膜缺陷, 载流子非辐射复合过程得到了抑制. 此外, 通过时间分辨荧光光谱(TRPL)进一步验证钙钛矿薄膜质量. 如图6(b)所示, FTO/CsPbIBr2结构下3个样品的平均衰减时间由0.76 ns提高至1.42 ns和1.60 ns, 说明前驱体膜处理后制备的钙钛矿薄膜有更少的缺陷, 载流子分离和传输效率更高.
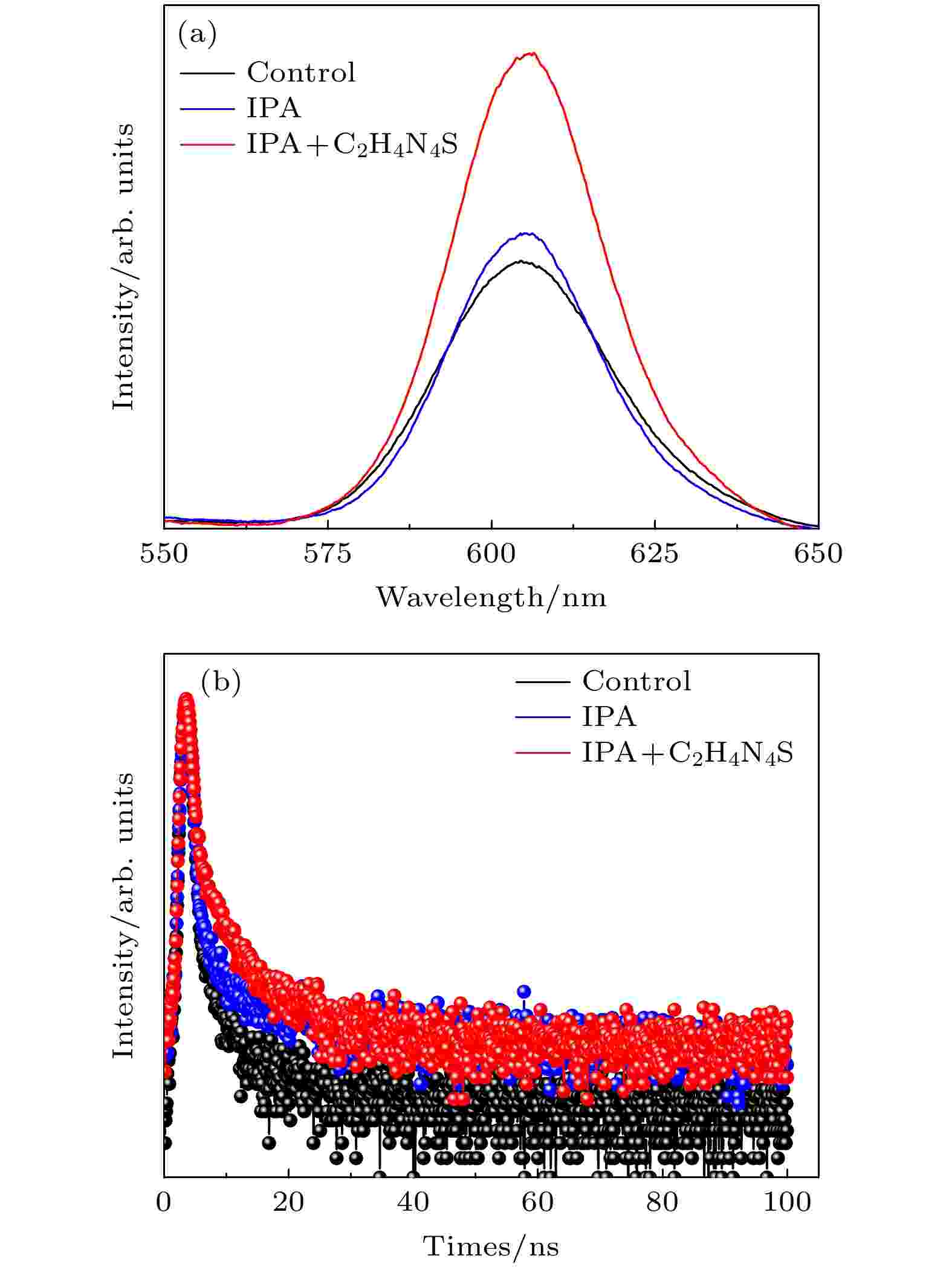 图 6 不同制备工艺下CsPbIBr2薄膜 (a) PL谱图; (b) TRPL谱图
图 6 不同制备工艺下CsPbIBr2薄膜 (a) PL谱图; (b) TRPL谱图Figure6. CsPbIBr2 film under different preparation processes: (a) PL spectra; (b) TRPL spectra.
为了探究前驱体薄膜处理工艺对于光伏器件性能的影响, CsPbIBr2太阳能电池的J-V性能曲线如图7(a)所示, 不同制备方法以及不同钝化剂浓度下器件性能具体参数在表1进行了总结. 通过图表可以发现, 参比器件中由于CsPbIBr2薄膜孔洞较多, 导致部分碳电极与SnO2电子传输层直接接触, 造成了载流子的非辐射复合, 而经过IPA及C2H4N4S处理后的薄膜致密性和平滑度都有显著改善. 当C2H4N4S浓度为0.4 mg/mL时, SCN–与Pb2+的配位钝化效果达到最佳, 有效降低了钙钛矿薄膜的缺陷, 此时器件光电转换效率达到最高的6.71%, 与参比器件5.29%的效率相比提升了近21.16%. 随着C2H4N4S浓度的进一步提高, 过多溶质在CsPbIBr2薄膜表面富集会提高薄膜表面粗糙度, 阻碍了载流子的传输, 导致电池光电性能下降.
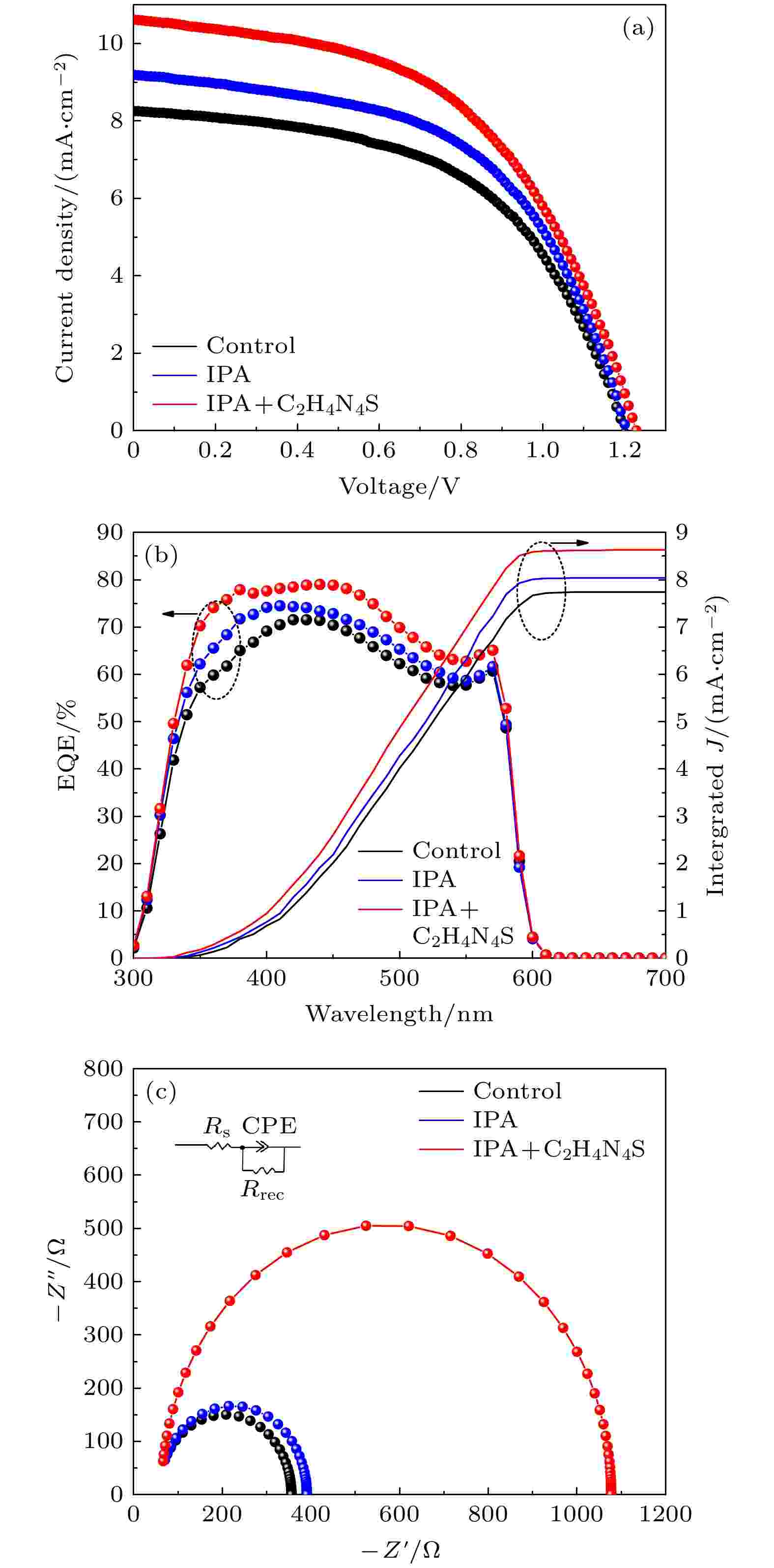 图 7 不同制备工艺下器件 (a) J-V曲线; (b) 外量子效率(external quantum efficiency, EQE)和积分电流曲线; (c) 电化学阻抗谱
图 7 不同制备工艺下器件 (a) J-V曲线; (b) 外量子效率(external quantum efficiency, EQE)和积分电流曲线; (c) 电化学阻抗谱Figure7. PSCs under different preparation processes: (a) J-V curves; (b) EQE spectra along with integrated current densities; (c) Nyquist plots.
| Perovskite | Jsc/mA cm–2 | Voc/V | FF/% | PCE/% |
| Control | 8.25 | 1.19 | 53.52 | 5.29 |
| IPA | 9.19 | 1.21 | 53.77 | 5.95 |
| IPA+0.2 mg/mL C2H4N4S | 10.32 | 1.21 | 50.10 | 6.24 |
| IPA+0.4 mg/mL C2H4N4S | 10.61 | 1.23 | 52.40 | 6.71 |
| IPA+0.6 mg/mL C2H4N4S | 10.89 | 1.19 | 50.12 | 6.38 |
| IPA+0.8 mg/mL C2H4N4S | 10.30 | 1.19 | 49.42 | 6.07 |
| 注: FF即填充因子(fill factor); PCE即光电转换效率(photoelectric conversion efficiency). | ||||
表1不同C2H4N4S浓度下钙钛矿电池性能指标
Table1.Photovoltaic Parameters of PSCs based on different concentration of C2H4N4S
图7(b)展示了3种制备方法下样品的EQE谱图. 经前驱体膜工艺处理后, 由于薄膜结晶质量得到改善, 样品在300—600 nm的波长范围内吸光能力有明显增强, 积分电流由7.74 mA/cm2提升至8.04 mA/cm2和8.63 mA/cm2, 说明钙钛矿太阳能器件的光电转换能力有所提高. 器件的Nyquist谱图如图7(c)所示, 包括器件等效电路图以及拟合曲线. 图中对比可以看到载流子的复合电阻(Rrec)分别为390.7 Ω和1077.1 Ω, 高于参比器件的355.8 Ω, 说明经前驱体膜工艺处理后器件的非辐射复合现象得到抑制, 这也是优化后器件中Voc有所提升的原因.
将钙钛矿电池在未封装条件下至于相对湿度为20%、室温25 °C避光环境中, 测试器件的稳定性, 实验结果如图8所示. 可以看出, 参比电池效率衰减明显在20天后效率只有初始的67.2%, 而IPA反溶及C2H4N4S钝化后的电池效率仍可保持70.5%和78.6%. 说明吸光层经前驱体膜处理工艺后能够有效提高钙钛矿电池的稳定性. 这是由于钙钛矿薄膜中的缺陷可以作为氧气和水分子的传输通道以促进离子的迁移. 而C2H4N4S与CsPbIBr2的相互作用能够调节薄膜表面形貌并形成疏水保护层, 抑制了钙钛矿材料的降解. 此外, C2H4N4S的加入加强了钙钛矿中Pb2+的螯合作用, 进一步钝化了薄膜整体缺陷, 从而提高了器件环境稳定性.
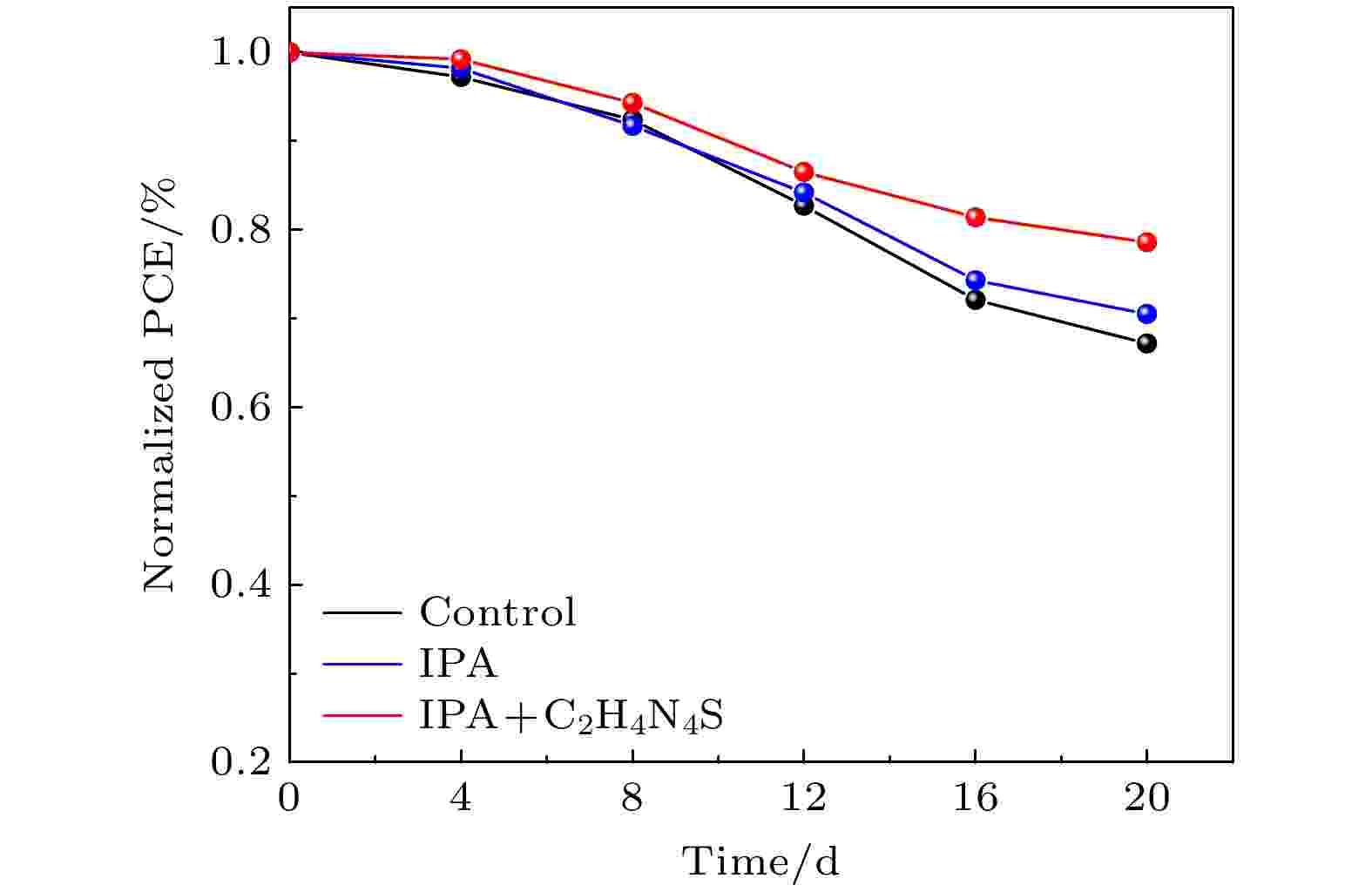 图 8 不同制备工艺下器件稳定性测试
图 8 不同制备工艺下器件稳定性测试Figure8. Stability of PSCs under different preparation processes.
