摘要/Abstract
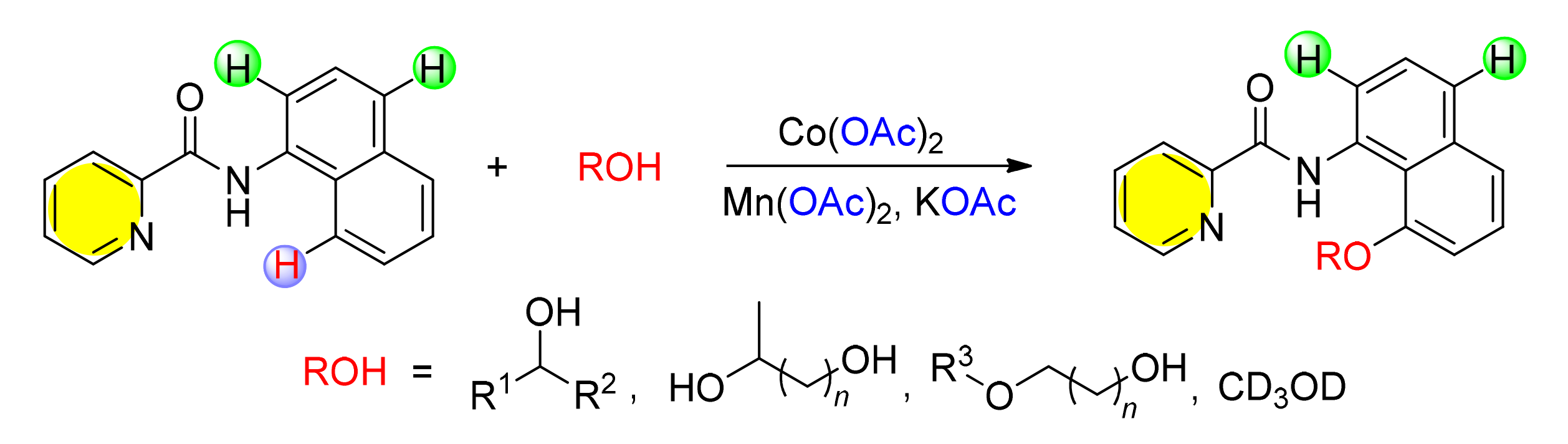
研究了吡啶酰胺双齿导向的钴催化1-萘胺衍生物的区域选择性碳氢键烷氧基化反应.研究发现不仅一元醇可以作为烷氧化剂在标准条件下较好地实现1-萘胺C(8)位的烷氧化反应,而且具有多重用途的脂肪二元醇以及低聚乙二醇,也可以以中等的收率得到相应的目标化合物,这可能是钴催化碳氢键活化构筑碳氧键的首次发现.此外,利用这个实验方法,以氘代甲醇为烷氧化试剂实现了同位素标记的8-烷氧基取代的1-萘胺衍生物的合成.通过控制实验,发现该反应中吡啶酰基是最佳的双导向基团,而且反应过程可能经历了单电子转移机理.
关键词: 钴催化, 双导向, 1-萘胺, C (8)-烷氧化, 区域选择性
The cobalt-catalyzed regioselective C-H alkoxylation of 1-naphthylamide with alcohols through a bidentate-chelation assistance has been developed. In this transformation, not only primary and secondary alcohols, but also aliphatic diols and oligoethylene glycols, which always be employed as O,O-donor ligands and reducing agents in transition metal catalyzed coupling reaction, were all tolerated under current reaction conditions. It is noteworthy that deuterium labeled 8-alkoxyl-1-N-(naphthalen-1-yl)picolinamide derivative was easily achieved under this catalytic system. In addition, control experiments suggested that picolinoyl was the key directing group, and furthermore, the C(8)-H alkoxylation reaction might proceed through a single-electron-transfer (SET) process.
Key words: cobalt-catalyzed, bidentate-assisted, 1-naphthylamide, C (8)-alkoxylation, regioselectivity
PDF全文下载地址:
点我下载PDF
