 ,中国农业科学院农业资源与农业区划研究所,北京100081
,中国农业科学院农业资源与农业区划研究所,北京100081Noncontact Inhibitory of Volatile Organic Compounds from Rice Root Bacteria on Rhizopus microsporus
WANG EnZhao, FAN FenLiang, LI YanLing, LIU XiongDuo, LU YuQiu, SONG ALin ,Institute of Agricultural Resources and Regional Planning, Chinese Academy of Agricultural Sciences, Beijing 100081
,Institute of Agricultural Resources and Regional Planning, Chinese Academy of Agricultural Sciences, Beijing 100081通讯作者:
责任编辑: 岳梅
收稿日期:2019-09-27接受日期:2019-11-7网络出版日期:2020-05-16
| 基金资助: |
Received:2019-09-27Accepted:2019-11-7Online:2020-05-16
作者简介 About authors
王恩召,E-mail:enzhaowang@163.com。

摘要
关键词:
Abstract
Keywords:
PDF (1380KB)元数据多维度评价相关文章导出EndNote|Ris|Bibtex收藏本文
本文引用格式
王恩召, 范分良, 李艳玲, 刘雄舵, 卢玉秋, 宋阿琳. 水稻根系细菌挥发性有机物对小孢根霉的非接触性抑制作用[J]. 中国农业科学, 2020, 53(10): 1986-1996 doi:10.3864/j.issn.0578-1752.2020.10.006
WANG EnZhao, FAN FenLiang, LI YanLing, LIU XiongDuo, LU YuQiu, SONG ALin.
0 引言
【研究意义】水稻在生长过程中根系能够释放各种信号分子,组装特有的根系微生物群落[1]。水稻根系与微生物通过密切的联系来实现互惠互利,微生物能将一些关键营养元素转化为更有利于植物吸收的形式,同时,水稻根系分泌物为微生物生长提供必要的营养元素[2,3]。微生物也能够释放各种代谢产物,如植物激素、挥发性有机物(microbial volatile organic compounds,mVOCs)等,它们在水稻生长发育过程中起着重要作用,不仅能促进植物生长,也能帮助植物抵御各种胁迫、病害等[4,5]。挥发性有机物是一类具有低分子量、高蒸气压、低水溶性特点的物质,在20℃、0.01 kPa下能快速挥发,进入气相状态[6]。植物可以通过释放挥发性有机物来达到信息传递和相互作用的目的,同样,微生物也能产生大量种类丰富、功能多样的挥发性物质[7]。微生物挥发性有机物主要包含烯烃、醇、酮、萜、苯、吡嗪、酸、酯、胺、醛类等物质,它们在农业生产过程中发挥着重要作用[8,9,10,11]。因此,研究微生物挥发性有机物对于调节农业生产,实现农业绿色可持续发展具有重要意义。【前人研究进展】据文献报道,微生物通过释放的挥发性有机物,不仅能够影响植物的生长发育,而且能够在非接触情况下,调节植物所面临的各种生物胁迫与非生物胁迫[12,13,14]。例如,LEE等[15]将植物置于木霉菌株释放的挥发性有机物中,植物的生物量和叶绿素含量显著增加,表明木霉菌株产生了促进植物生长的挥发性有机物,但也有部分木霉释放的挥发性有机物降低了植物生长量和叶绿素含量,表明不同种类的菌株释放的挥发性有机物对植物生长的作用不同;CHOUDOIR等[16]从土壤和空气的尘埃中分离出48种放线菌,并检测了这些放线菌在不同培养基上释放的挥发性有机物,发现每个菌株都产生独特的挥发性有机物组合,尽管某些化合物是由许多菌株产生的,但大多数是菌株特异性的。重要的是,密切相关的菌株之间的挥发性有机化合物特征更为相似,通过检测这些挥发性有机物对致病性和非致病性假单胞菌(Pseudomonas)菌株生长速率的影响,发现其对代表植物有益微生物和致病微生物的生长具有刺激和抑制作用;XU等[17]使用固相微萃取-气相色谱/质谱法鉴定了部分土壤中微生物挥发性有机化合物,并采用相对纯商业化合物进行体外拮抗试验,发现即使在低浓度下三甲胺、苯甲醛和N,N-二甲基辛胺也具有较强的抗真菌活性。越来越多证据表明,微生物挥发性有机物对生态环境友好,可以作为一种经济有效的可持续战略应用于农业实践。【本研究切入点】不同植物的根际微生物往往与众不同,每种微生物又能产生独特的挥发性有机物,前期研究大多集中在微生物通过释放挥发性有机物影响植物或微生物生长[18,19,20,21],而对水稻生长过程根系微生物挥发性有机物研究较少。【拟解决的关键问题】筛选水稻根系能够通过释放挥发性有机物来抑制病原真菌小孢根霉(Rhizopus microsporus)生长的细菌,并对具有不同抑菌效果的细菌释放的挥发性有机物进行检测,确定具有抑菌活性的物质并采用相应的纯化学品进行验证,以期鉴定出新的具有潜在抑菌活性的挥发性有机物。1 材料与方法
试验于2018年6月至2019年7月在中国农业科学院农业资源与农业区划研究所完成。1.1 供试菌株、培养基、培养条件
9种供试菌株耳炎假单胞菌(Pseudomonas otitidis)(编号为3)、烟草肠杆菌(Enterobacter tabaci)(编号为26)、神户肠杆菌(Enterobacter kobei)(编号为34)、假单胞菌(Pseudomonas sp.)(编号为35)、皮氏罗尔斯通氏菌(Ralstonia pickettii)(编号为50)、阿氏芽孢杆菌(Bacillus aryabhattai)(编号为62,该菌株已申请专利,专利申请号:2019108367651)、节杆菌(Arthrobacter sp.)(编号为146)、短短芽孢杆菌(Brevibacillus brevis)(编号为2-18)、麦克默多生孢八叠球菌(Paenisporosarcina macmurdoensis)(编号为2-60)由笔者从水稻根系筛选,均能通过释放挥发性有机物对小孢根霉生长产生抑制作用。冷冻保藏的菌株采用牛肉膏蛋白胨培养基进行活化(牛肉膏3 g,蛋白胨10 g,氯化钠5 g,琼脂20 g,蒸馏水1 L,pH 7.2—7.4),黑暗条件、30℃恒温培养48 h。将活化好的菌株接种于液体的牛肉膏蛋白胨培养基(不加琼脂)中,180 r/min、30℃振荡培养48 h后获得菌株发酵液,备用。小孢根霉由笔者实验室在水稻根际分离、鉴定、保存。活化小孢根霉采用PDA培养基(马铃薯200 g,葡萄糖20 g,琼脂20 g,水1 L,自然pH),黑暗条件、30℃恒温培养48 h。将活化好的菌株接种于PDB培养基(配方同PDA,不加琼脂)中,180 r/min、30℃振荡培养48 h后获得菌株发酵液,备用。
1.2 细菌挥发性有机物对小孢根霉的抑制作用
取供试菌株菌液100 μL至牛肉膏蛋白胨培养基上,用121℃高压蒸汽灭过菌的涂布棒涂均匀,放到30℃培养箱培养24 h。取小孢根霉发酵液10 μL,滴在PDA培养基中央,然后将培养24 h的供试菌株培养皿扣在其上,对照为将不加菌的牛肉膏蛋白胨培养基扣上,周围用封口膜密封,黑暗条件下30℃恒温培养,每隔24 h观察记录一次,直到小孢根霉长满板,培养结束,每处理4个重复,比较挥发性有机物的抑菌效果。抑菌率=(对照小孢根霉菌落直径-处理小孢根霉菌落直径)/对照小孢根霉菌落直径×100%。1.3 挥发性有机物的收集
取5 mL牛肉膏蛋白胨培养基于20 mL顶空瓶中,加入0.1 g琼脂,用牛皮纸封上瓶口,121℃、30 min灭菌,灭菌完将瓶子倾斜30°,冷却凝固,取100 μL预先准备好的菌液加入瓶中,摇晃瓶体使菌液均匀分布在培养基斜面上,盖上牛皮纸,黑暗条件、30℃恒温培养48 h,去掉牛皮纸,用带有聚四氟乙烯隔垫的中空螺旋盖封口,同时设置只加培养基、不接菌的顶空瓶作为空白对照,每处理4个重复。培养5 d后,采用50/30 μm DVB/CAR/PDMS萃取纤维(购于Supelco公司)进行萃取,萃取头装配于SPME手动萃取手柄上(Supelco,Bellefonte,PA,USA),在使用前先进行老化,老化按厂家说明书进行,之后在30℃恒温条件下萃取12 h。
1.4 挥发性有机物的分析
萃取完成后立即采用安捷伦7890A-5975C气相色谱-质谱分析仪(Agilent Technologies,USA)进行检测分析。色谱条件:进样口温度250℃,进样时间3 min;不分流模式,载气为99.999%的高纯氦气,柱流速为1 mL·min-1。柱箱升温程序:初始温度50℃,保持2 min,以8℃·min-1的速度升温至180℃,再以10℃·min-1的速度升温至240℃,保持6 min。质谱条件:离子化方式为EI,70 eV;离子源温度为230℃,四级杆温度150℃,传输线温度250℃;扫描模式为全扫描,扫描范围35—450 amu。测得的挥发性有机物质谱在NIST/EPA/NIH数据库中进行比对鉴定。1.5 纯品体外拮抗验证
在PDA培养基中加入一个灭过菌的2 mL离心管盖子,盖子里分别加入100 μL的3-甲基丁酸和5-甲基-2-己酮,对照组盖子里加入100 μL灭菌水,培养基中央加入10 μL小孢根霉菌液,32℃黑暗条件下培养,观察小孢根霉生长状况。2 结果
2.1 挥发性有机物对小孢根霉的抑制作用
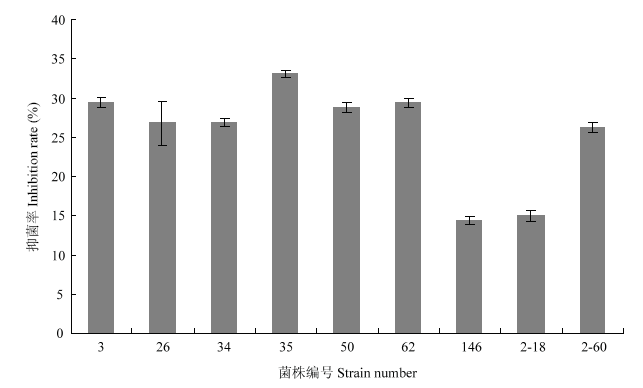
9株供试细菌菌株分别对小孢根霉产生了不同程度的抑制作用,3、26、34、35、50、62和2-60号菌株的抑菌率相差不大,分别为29.38%、26.88%、26.88%、33.13%、28.75%、29.38%和26.25%,抑菌率相对高于菌株146(14.38%)和2-18(15.00%)号(图1)。图1
 新窗口打开|下载原图ZIP|生成PPT
新窗口打开|下载原图ZIP|生成PPT图1不同细菌对小孢根霉的抑菌率
Fig. 1Inhibition rate of different bacteria against R. microsporus
2.2 供试菌株释放挥发性有机物的定性分析
每个供试菌株都产生了不同种类的挥发性有机物,这些物质有菌株独有的,也有共同产生的(表1),且每种菌产生的挥发性有机物数量以及浓度也不同,其中3号菌释放7种物质、26号菌释放17种物质、34号菌释放21种物质、35号菌释放30种物质、50号菌释放10种物质、62号菌释放29种物质、146号菌释放10种物质、2-18号菌释放18种物质、2-60号菌释放33种物质。Table 1
表1
表1微生物挥发性有机化合物的组成
Table 1
| 挥发性有机物名称VOCs name | 2-60 | 34 | 35 | 62 | 146 | 2-18 | 26 | 50 | 3 |
|---|---|---|---|---|---|---|---|---|---|
| (E)-1-甲基-2-(丙-1-烯-1-基)二硫 (E)-1-Methyl-2-(prop-1-en-1-yl) disulfane | - | - | - | - | - | - | + | - | - |
| β-苯基乙基丁酸酯beta-Phenylethyl butyrate | - | - | - | - | - | - | + | - | - |
| 11-十二烯-2-酮11-Dodecen-2-one | - | - | - | - | - | + | - | - | - |
| 2-甲基-1-丁醇2-Methyl-1-butanol | - | + | - | - | - | - | - | - | - |
| 3-甲基-1-丁醇3-Methyl-1-butanol | + | + | + | + | + | - | + | - | - |
| 1-丁醇,3-甲基-乙酸1-Butanol, 3-methyl-acetate | - | + | + | - | - | - | - | - | - |
| 2-乙基-1-己醇2-Ethyl-1-hexanol | + | - | + | + | + | + | - | - | + |
| 1-十八碳烯1-Octadecene | + | - | - | - | - | - | - | - | - |
| 1-辛醇1-Octanol | + | - | - | - | - | - | - | - | - |
| 7-甲基-1-辛烯7-Methyl-1-octene | + | - | - | - | - | - | - | - | - |
| 1-十五碳烯1-Pentadecene | - | - | - | - | - | - | + | - | - |
| 2-氯丙酸,十六烷基酯2- Chloropropionic acid, hexadecyl ester | - | - | - | - | - | - | + | - | - |
| 5-庚基二氢-2(3H)-呋喃酮5-Heptyldihydro-2(3H)-furanone | - | - | + | - | + | - | - | - | - |
| 2,4,6-环庚三烯-1-酮2,4,6-Cycloheptatrien-1-one | + | - | - | - | - | - | - | - | - |
| 2,4-二硫戊烷2,4-Dithiapentane | - | - | - | - | - | - | - | - | + |
| 6-甲基-2,4-庚二酮6-Methyl-2,4-heptanedione | - | - | - | - | - | + | - | - | - |
| 3,7,11-三甲基-2,6,10-十二碳三烯-1-醇 3,7,11-Trimethyl-2,6,10-dodecatrien-1-ol | - | - | - | - | - | - | - | - | + |
| 2-癸酮2-Decanone | + | + | + | + | - | + | + | - | - |
| 2-十二烷酮2-Dodecanone | - | - | + | - | - | + | - | - | - |
| 2-乙基-1-己醇2-Ethyl-1-hexanol | - | - | - | - | - | - | - | + | - |
| 2-庚酮2-Heptanone | + | + | + | + | - | + | + | - | - |
| 3-甲基-2-庚酮3-Methyl-2-heptanone | - | - | - | + | - | - | - | - | - |
| 5-甲基-2-庚酮5-Methyl-2-heptanone | + | - | - | + | - | + | - | - | - |
| 6-甲基-2-庚酮6-Methyl-2-heptanone | + | - | + | + | - | + | - | - | - |
| 2-十六烷酮2-Hexadecanone | - | - | - | - | - | + | - | - | - |
| 2-己酮2-Hexanone | + | - | + | + | - | - | - | - | - |
| 3,4-二甲基-2-己酮3,4-Dimethyl-2-hexanone | + | - | - | - | - | - | - | - | - |
| 5-甲基-2-己酮5-Methyl-2-hexanone | + | - | - | + | - | + | - | - | - |
| 2-壬基酮2-Nonanone | - | + | + | + | - | + | + | - | - |
| 2-辛酮2-Octanone | - | - | + | + | - | - | - | - | - |
| 2-十五烷酮2-Pentadecanone | - | + | - | - | - | - | - | - | - |
| 2-戊酮2-Pentanone | - | + | + | - | - | - | - | - | - |
| 3-甲基-2-戊酮3-Methyl-2-pentanone | - | - | - | - | - | + | - | - | - |
| 2-十四烷酮2-Tetradecanone | - | - | + | + | - | + | - | - | - |
| 2-十三烷酮2-Tridecanone | - | - | - | + | - | - | - | - | - |
| 2-十一烷酮2-Undecanone | - | + | + | - | - | + | + | - | - |
| 3-十二烷酮3-Dodecanone | - | - | - | + | - | - | - | - | - |
| 7-苯基-3-庚烯7-Phenyl-3-heptene | - | + | - | - | - | - | - | - | - |
| 5-甲基-3-己酮5-Methyl-3-hexanone | + | - | + | + | - | - | - | - | - |
| 3-十五烷酮3-Pentadecanone | - | - | - | + | - | - | - | - | - |
| 3-戊酮3-Pentanone | - | - | - | + | - | - | - | - | - |
| 3-十三烷酮3-Tridecanone | + | - | - | - | - | + | - | - | - |
| 5-甲基-4-己烯-3-酮5-Methyl-4-hexen-3-one | + | - | - | - | - | - | - | - | - |
| 6,10,14-三甲基-5,9,13-戊三烯-2-酮6,10,14-Trimethyl-5,9,13-pentadecatrien-2-one | - | - | - | - | - | - | - | + | - |
| 6-甲基-5-庚烯-2-酮6-Methyl-5-hepten-2-one | + | - | - | - | - | - | - | + | - |
| 6-叔丁基-2,4-二甲基苯酚6-Tert-butyl-2,4-dimethylphenol | - | + | + | - | - | - | - | - | - |
| 7-甲基辛烷-2,4-二酮,烯醇式7-Methyloctane-2,4-dione, enol form | - | - | - | - | - | + | - | - | - |
| 乙酸,2-苯乙基酯Acetic acid, 2-phenylethyl ester | - | + | - | - | - | - | - | - | - |
| 乙酸,氯-十六烷基酯Acetic acid, chloro-hexadecyl ester | - | + | - | - | - | - | - | - | - |
| 顺式乙酸,非3-烯基酯cis-Acetic acid, non-3-enyl ester | - | - | + | - | - | - | - | - | - |
| (2-甲氧基乙基)-苯 (2-Methoxyethyl)-benzene | - | - | - | - | - | - | + | - | - |
| (甲氧基甲基)-苯 (Methoxymethyl)-benzene | - | - | - | - | - | - | + | - | - |
| 苯乙酸,乙酯Benzeneacetic acid, ethyl ester | + | - | - | - | - | - | - | - | - |
| 苯甲酸,乙酯Benzoic acid, ethyl ester | - | - | - | - | - | - | - | + | - |
| 苯甲醇Benzyl alcohol | - | - | - | + | + | - | - | - | - |
| 苄基甲基酮Benzyl methyl ketone | + | - | - | + | + | + | + | - | - |
| 1-甲氧基-3-甲基丁烷1-Methoxy-3-methyl-butane | - | - | - | - | - | - | + | - | - |
| 2-甲基-2-(甲硫基)-丁烷2-Methyl-2-(methylthio)-butane | - | - | - | - | - | - | - | + | - |
| 丁烷磺酸,硫-甲酯Butanethioic acid, S-methyl ester | + | - | - | - | - | - | - | - | - |
| 丁酸,1-乙烯基己酯Butanoic acid, 1-ethenylhexyl ester | - | - | + | - | - | - | - | - | - |
| 3-甲基丁酸3-Methyl-butanoic acid | - | + | + | + | - | - | - | - | - |
| 丁酸,3-甲基-乙酯Butanoic acid, 3-methyl-ethyl ester | + | - | - | + | - | - | - | - | - |
| 顺式双环[3.3.0]辛-2-烯cis-Bicyclo[3.3.0]oct-2-ene | + | - | - | - | - | - | - | - | - |
| 环庚烯Cycloheptene | - | - | - | - | - | - | + | - | - |
| 1-甲基环己烯1-Methyl-cyclohexene | - | - | - | + | - | - | - | - | - |
| 3-乙烯基-环戊烯3-Ethenyl-cyclopentene | + | - | - | - | - | - | - | - | - |
| 双环戊二烯Dicyclopentadiene | + | + | + | + | - | + | - | + | - |
| 三硫化二甲基Dimethyl trisulfide | + | + | + | + | + | - | - | + | + |
| 二硫化二甲基Dimethyl disulfide | + | + | + | + | + | - | + | + | + |
| 十二酸,乙酯Dodecanoic acid, ethyl ester | + | - | - | - | - | - | - | - | - |
| 1-(2-氨基苯基)-乙酮1-(2-Aminophenyl)-ethanone | - | + | - | - | - | - | + | - | - |
| 13-甲基十四烷酸乙酯Ethyl 13-methyl-tetradecanoate | + | - | - | - | - | - | - | - | - |
| 3-(甲硫基)-(E)-2-丙酸乙酯Ethyl 3-(methylthio)-(E)-2-propenoate | - | - | - | - | - | - | - | + | - |
| 3-(甲硫基)-(Z)-2-丙酸乙酯Ethyl 3-(methylthio)-(Z)-2-propenoate | - | - | - | - | - | - | - | + | - |
| 十三烷酸乙酯Ethyl tridecanoate | + | - | - | - | - | - | - | - | - |
| 甲基异丁基酮Methyl isobutyl ketone | + | - | + | + | - | - | - | - | - |
| 异戊酸甲酯Methyl isovalerate | - | - | + | - | - | - | - | - | - |
| 硫羟乙酸甲酯Methyl thiolacetate | - | - | - | - | - | - | - | - | + |
| 十氢萘Decahydro-naphthalene | - | + | + | - | - | - | - | - | - |
| 十八烷Octadecanal | - | - | + | - | - | - | - | - | - |
| 苯乙醇Phenylethyl alcohol | - | + | + | + | - | - | + | - | - |
| 丙酸,2-苯乙基酯Propanoic acid, 2-phenylethyl ester | - | + | - | - | - | - | - | - | - |
| 2-乙基-5-甲基吡嗪2-Ethyl-5-methyl-pyrazine | - | - | + | - | - | - | - | - | - |
| 三甲基吡嗪Trimethyl-pyrazine | - | - | + | - | - | - | - | - | - |
| 吡咯Pyrrole | - | - | - | + | - | - | - | - | - |
| 3-甲基丁硫醇甲酯S-Methyl 3-methylbutanethioate | + | - | + | + | + | - | - | - | + |
| 丙酮三氧化物TATP | - | - | - | - | + | - | - | - | - |
| 十四烷酸,乙酯Tetradecanoic acid, ethyl ester | + | - | - | - | - | - | - | - | - |
| 2-甲氧基-5-甲基-噻吩2-Methoxy-5-methyl-thiophene | - | - | - | - | + | - | - | - | - |
| 十一酸,乙酯Undecanoic acid, ethyl ester | + | - | - | - | - | - | - | - | - |
新窗口打开|下载CSV
2.3 挥发性有机物的定量分析
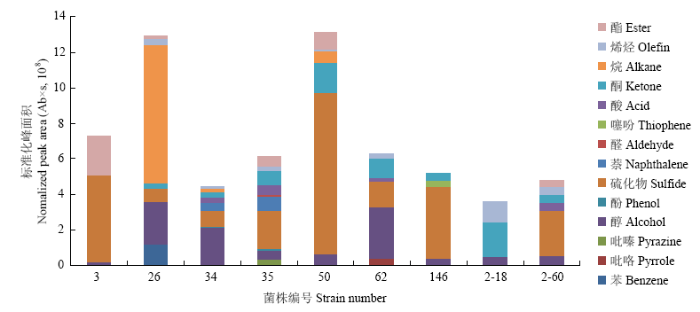
供试菌株释放的挥发性有机物主要包括酯、烯烃、烷、酮、酸、噻吩、醛、萘、硫化物、酚、醇、吡嗪、吡咯、苯类,其中所有菌株都释放醇类物质,3、35、50、146、2-60号菌株释放的硫化物占比最大,26号菌株释放的烷类物质占比最大,34、62号菌株释放的醇类物质占比最大,2-18号菌株释放的酮类物质占比最大(图2)。图2
 新窗口打开|下载原图ZIP|生成PPT
新窗口打开|下载原图ZIP|生成PPT图2不同细菌释放的挥发性有机物组成
Fig. 2The composition of VOCs emitted from different bacteria
2.4 挥发性有机物的数量组成分析
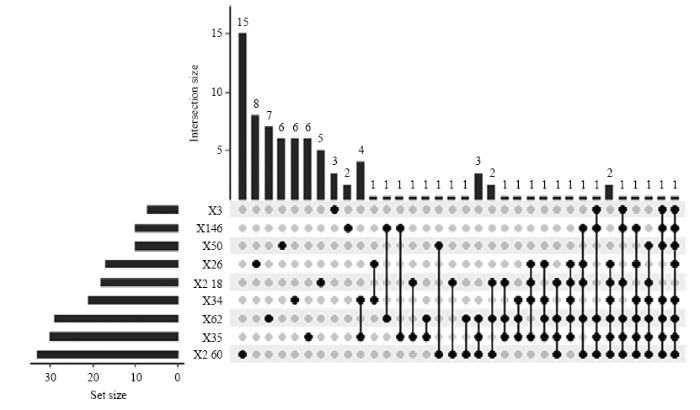
所有供试菌株共检测到90种物质,虽然每种菌都能释放自己独特的挥发性有机物,但是它们释放的物质也存在着一定的交集,2-60号菌独有的物质有15种,35号独有6种,62号独有7种,34号独有6种,2-18号独有5种,26号独有8种,50号独有6种,146号独有2种,3号独有3种。其中8种菌同时释放的有1种物质,7种菌同时释放的有1种物质,6种菌同时释放的有5种物质,5种菌同时释放的有3种物质,4种菌同时释放的有3种物质,3种菌同时释放的有7种物质,其余为2种菌同时释放的物质(图3)。图3
 新窗口打开|下载原图ZIP|生成PPT
新窗口打开|下载原图ZIP|生成PPT图3不同菌株释放不同和相同挥发性有机物数量的韦恩图
左侧显示为每个菌株释放的挥发性有机物总数,上面显示为每个菌株独有或与其他菌株共有的挥发性有机物数量
Fig. 3The Venn diagram showing the amount of different and same VOCs released by different strains
The left side shows the total amount of VOCs released by each strain, and the above shows the amount of VOCs unique to each strain or shared with other strains
2.5 挥发性有机物与抑菌率之间的关系
通过相关性分析,发现有6种物质(2-庚酮、5-甲基-2-己酮、2-壬酮、5-甲基-3-己酮、3-甲基丁酸和甲基异丁基酮)在质谱上的峰面积和相对应菌株的抑菌率呈显著正相关,其中有3种物质(5-甲基-2-己酮、5-甲基-3-己酮和3-甲基丁酸)文献中暂未报道(表2)。Table 2
表2
表2细菌挥发性有机化合物质谱上的峰面积与相应菌株抑菌率的相关性系数
Table 2
| 挥发性有机物名称 VOCs name | 相关性系数 Correlation coefficient | 挥发性有机物名称 VOCs name | 相关性系数 Correlation coefficient |
|---|---|---|---|
| 3-甲基-1-丁醇3-Methyl-1-butanol (+) | 0.093 | 2-十一烷酮2-Undecanone (+) | 0.485 |
| 2-乙基-1-己醇2-Ethyl-1-hexanol | -0.396 | 5-甲基-3-己酮5-Methyl-3-hexanone (++) | 0.863** |
| 2-癸酮2-Decanone | 0.074 | 苄基甲基酮Benzyl methyl ketone | -0.169 |
| 2-庚酮2-Heptanone (+) | 0.544** | 3-甲基丁酸3-Methyl-butanoic acid (++) | 0.691* |
| 5-甲基-2-庚酮5-Methyl-2-heptanone | -0.705* | 双环戊二烯Dicyclopentadiene | 0.025 |
| 6-甲基-2-庚酮6-Methyl-2-heptanone | 0.146 | 三硫化二甲基Dimethyl trisulfide (+) | -0.494* |
| 2-己酮2-Hexanone | -0.251 | 二硫化二甲基Dimethyl disulfide (+) | -0.074 |
| 5-甲基-2-己酮5-Methyl-2-hexanone (++) | 0.710** | 甲基异丁基酮Methyl isobutyl ketone (+) | 0.781** |
| 2-壬酮2-Nonanone (+) | 0.514* | 苯乙醇Phenylethyl alcohol | -0.576* |
| 2-十四烷酮2-Tetradecanone | -0.882** | 3-甲基丁硫醇甲酯S-Methyl 3-methylbutanethioate | 0.346 |
新窗口打开|下载CSV
2.6 纯品体外检测
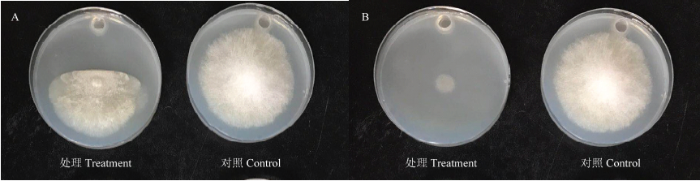
选取能购买到的其中2种物质(5-甲基-2-己酮和3-甲基丁酸)做体外验证,发现3-甲基丁酸严重影响小孢根霉生长,5-甲基-2-己酮对小孢根霉具有致死作用(图4)。图4
 新窗口打开|下载原图ZIP|生成PPT
新窗口打开|下载原图ZIP|生成PPT图43-甲基丁酸(A)和5-甲基-2-己酮(B)熏蒸小孢根霉
对照为无菌水熏蒸小孢根霉
Fig. 4R. microspores treated with 3-methyl-butanoic acid (A) and 5-methyl-2-hexanone (B)
The control is treated with sterile water
3 讨论
大量文献报道细菌能通过释放挥发性有机化合物来抑制病原菌的生长[22,23],王静等[24]在烟草根际分离到一株短小芽孢杆菌(Bacillus pumilus)AR03,该菌株能产生多种挥发性有机物,且挥发物对烟草黑胫病菌(Phytophthora parasitica var. nicotianae)和赤星病菌(Alternaria alternata)均具有抑制作用。本研究通过对扣熏蒸法,发现有9株细菌能够在不接触小孢根霉的情况下抑制其生长,并且抑制效果不同(图1),说明这些细菌释放了能够抑制病原菌生长的挥发性物质,虽然文献里有较多相关报道,但对于水稻根系细菌释放的挥发性有机物却鲜有报道。通过对本研究的9株细菌释放的挥发性有机物进行检测,发现这些细菌每株都释放了多种挥发性有机物,每株菌释放的挥发性有机物有重叠的部分,也都特有部分,同时,每种挥发性有机物的量也不同(表1、图2),研究结果与文献报道较为一致。如MACKIE等[21]随机选择土壤中细菌,测定对几种真菌生长的影响,发现54%菌株对部分真菌生长具有抑制作用;LI等[25]通过检测8株芽孢杆菌释放的挥发性有机物,发现这些菌株均能释放多种挥发性有机物,且含量不同。
本研究中,选取同时被3株以上菌株释放的物质,将它们在质谱上的峰面积与相对应菌株的抑菌率做相关性分析,发现有6种物质(2-庚酮、5-甲基-2-己酮、2-壬酮、5-甲基-3-己酮、3-甲基丁酸和甲基异丁基酮)与其对应菌株的抑菌率呈显著正相关(表2)。其中,有3种物质(2-庚酮、2-壬酮和甲基异丁基酮)在不同文献中被报道具有抑菌作用,如POPOVA等[26]从土壤或根际分离得到的假单胞菌和沙雷氏菌(Serratia)产挥发物对根癌农杆菌(Agrobacterium tumefaciens)和真菌具有抑制作用,对蓝藻、果蝇(黑腹果蝇Drosophila melanogaster)和线虫(秀丽隐杆线虫Caenorhabditis elegans)具有杀灭作用,通过检测发现主要是一些酮类物质(2-壬酮、2-庚酮和2-十一烷酮)和二甲基二硫起作用;EZRA等[27]研究内生真菌Muscodor albus的抑菌活性,通过检测其释放的挥发性有机物,发现该菌株在蔗糖培养基上主要释放甲基异丁基酮、乙酸和丁酯3种物质。本研究所筛选的6种挥发性有机物中尚有3种物质(5-甲基-2-己酮、5-甲基-3-己酮和3-甲基丁酸)暂未被报道具有抑菌作用,因此,选取能购买到的其中2种物质(5-甲基-2-己酮和3-甲基丁酸)进行体外熏蒸试验,发现5-甲基-2-己酮能够使小孢根霉致死,3-甲基丁酸严重抑制小孢根霉的生长(图4)。
另外,3-甲基-1-丁醇、2-十一烷酮、三硫化二甲基和二硫化二甲基这几种物质虽然本研究并未分析出具有抑菌活性,但有文献报道其具有抑菌活性[28]。如WANG等[29]通过分析一株白色链霉菌TD-1释放的挥发性有机物,发现该菌株释放最多的物质为二甲基二硫,在体外熏蒸试验中能抑制念珠菌的生长;FERNANDO等[30]通过检测从菜籽、大豆中分离出的细菌释放的挥发性有机物,发现6种物质(苯并噻唑、环己醇、正癸醛、三硫化二甲基、2-乙基1-己醇和壬醛)能抑制真菌生长。由于本研究是通过相关性分析来确定物质释放是否具有抑菌活性,因此笔者推测可能是释放这些物质的菌株同时释放了其他一些抑菌活性更强的物质,导致这些物质相对含量与菌株的抑菌率没有呈正相关,如50号菌株释放了文献已证明具有抑菌活性的物质三硫化二甲基和二硫化二甲基,虽然没有释放本研究中分析到的6种物质,但却具有一定的抑菌能力,也可能是小孢根霉对于这些物质的敏感性偏低,从而导致文献中提到的一些抑菌物质在本研究中未发现。
4 结论
水稻根系细菌能释放种类丰富的挥发性有机物,部分挥发性有机物对水稻病原菌小孢根霉具有显著抑制作用。通过相关性分析及体外验证确定了2种新型的具有抑菌活性的物质(5-甲基-2-己酮和3-甲基丁酸)以及1种可能具有抑菌活性的物质(5-甲基-3-己酮),这些菌株和挥发性有机物均具有作为新型药物和抗真菌代谢物生物资源的潜力。参考文献 原文顺序
文献年度倒序
文中引用次数倒序
被引期刊影响因子
DOI:10.1126/science.aat1168URL [本文引用: 1]
[本文引用: 1]
DOI:10.1038/s41564-018-0129-3URL [本文引用: 1]
DOI:10.1038/s41587-019-0104-4URL [本文引用: 1]
DOI:10.1038/s41396-018-0186-xURL [本文引用: 1]
DOI:10.1093/femsre/fuu013URL [本文引用: 1]
[本文引用: 1]
[本文引用: 1]
DOI:10.1016/j.fbr.2012.07.001URL [本文引用: 1]
DOI:10.1093/nar/gkt1250URL [本文引用: 1]
DOI:10.1093/femsec/fiw070URL [本文引用: 1]
DOI:10.1016/j.trac.2015.08.010URL [本文引用: 1]
DOI:10.1016/j.tplants.2015.01.004URL [本文引用: 1]
DOI:10.1016/j.tplants.2019.01.008URL [本文引用: 1]
DOI:10.1016/j.soilbio.2017.11.015URL [本文引用: 1]
DOI:10.1186/s40694-016-0025-7URL [本文引用: 1]
[本文引用: 1]
DOI:10.1016/j.soilbio.2004.07.020URL [本文引用: 1]
[本文引用: 1]
DOI:10.1016/j.ppees.2019.04.003URL [本文引用: 1]
[本文引用: 1]
DOI:10.1016/S0038-0717(98)00140-0URL [本文引用: 2]
DOI:10.1016/j.lwt.2016.12.020URL [本文引用: 1]
DOI:10.1007/s00253-016-7584-7URL [本文引用: 1]
DOI:10.3864/j.issn.0578-1752.2018.10.010URL [本文引用: 1]

【Objective】 The objective of this study is to determine the antimicrobial activity of volatile organic compounds (VOCs), produced from tobacco rhizosphere soil Bacillus pumilus AR03 strain, and to analyze its main components.【Method】 The antifungal effect of VOCs on the colony, mycelium growth and spore germination of Phytophthora parasitica var. nicotianae and Alternaria alternata was determined by a double Petri dish assay and cavity slide method. The control effect of VOCs on tobacco black shank and brown spot by leaf inoculation was determined in vitro. VOCs were collected by head-space solid phase microextraction (HS-SPME) and identified by gas chromatography-mass spectrometry (GC-MS). Retention index (RI) and internal standard (IS) 1-pentadecence were used for qualitative and quantitative analysis.【Result】 VOCs released from B. pumilus AR03 strain had certain inhibitory effect on the two target pathogens, which showed that the mycelium of P. parasitica grew slowly and thinned, branches increased and twisted, and broke. The mycelium of A. alternata was deformed and no conidial pedicle was produced on the mycelium. Most of the inclusions gathered together and caused the mycelium to dry and constricted. Growth of exposed fungus colonies was inhibited by VOCs, the inhibition rates of VOCs were 56.21% and 59.23%, 64.75% and 59.86%, 66.13% and 61.10%, 67.04% and 70.00%, respectively, against P. parasitica and A. alternata cultured in sealed plates for 2, 4, 6 and 8 d. When the zoospores of P. parasitica and ascospores of A. alternata exposed to these volatile components for 6 h, the germination was delayed and the growth was slow. The number of sporocyst produced by P. parasitica obviously reduced. Most conidiophores of A. alternata expanded abnormally as cystic structure, indicating the fungicidal nature of the volatiles. Moreover, VOCs could significantly inhibit the disease severities of tobacco black shank and brown spot on leaves tests. Direct fumigation for 40 h and 80 h, black shank disease incidence was 92.50% on control and 70.83% on leaves treated by VOCs, the inhibitory of spot expansion was 62.35%. Brown spot disease incidence was 88.33% on control and 60.80% on leaves treated by VOCs, lesions expanded slowly and inhibitory rate was 65.75%. SPME GC-MS analysis showed that seven components of the volatiles were identified, all of which are sesquiterpenes with C15H24 structure. They are dihydrocurcumene (CAS NO. 1461-02-5), (E)-β-famesene (CAS NO. 18794-84-8), γ-curcumene (CAS NO. 451-55-8), α-zingiberene (CAS NO. 495-60-3), π-bisabolene (CAS NO. 495-61-4), β-sesquiphellandrene (CAS NO. 20307-83-9) and γ-E-bisabolene (CAS NO. 53585-13-0). When AR03 was cultured for 1 d, the relative content of β-sesquiphellandrene was the highest (80.64%), followed by (E)-β-famesene and α-zingiberene, the relative content was 7.20% and 6.67%, respectively. With the extension of culturing time, the species of each component were the same, but the relative content was different. Except for dihydrocurcumene, the content of other components showed a decreasing trend, when cultured for 6 d, other ingredients decreased more than 50%, besides dihydrocurcumene keeping relatively stable. 【Conclusion】 VOCs produced by B. pumilus AR03 could develop an additive antifungal effect against fungal pathogens on tobacco. B. pumilus AR03 has potential as an important microbial resource for developing antifungal metabolites and new drugs.
DOI:10.3864/j.issn.0578-1752.2018.10.010URL [本文引用: 1]

【Objective】 The objective of this study is to determine the antimicrobial activity of volatile organic compounds (VOCs), produced from tobacco rhizosphere soil Bacillus pumilus AR03 strain, and to analyze its main components.【Method】 The antifungal effect of VOCs on the colony, mycelium growth and spore germination of Phytophthora parasitica var. nicotianae and Alternaria alternata was determined by a double Petri dish assay and cavity slide method. The control effect of VOCs on tobacco black shank and brown spot by leaf inoculation was determined in vitro. VOCs were collected by head-space solid phase microextraction (HS-SPME) and identified by gas chromatography-mass spectrometry (GC-MS). Retention index (RI) and internal standard (IS) 1-pentadecence were used for qualitative and quantitative analysis.【Result】 VOCs released from B. pumilus AR03 strain had certain inhibitory effect on the two target pathogens, which showed that the mycelium of P. parasitica grew slowly and thinned, branches increased and twisted, and broke. The mycelium of A. alternata was deformed and no conidial pedicle was produced on the mycelium. Most of the inclusions gathered together and caused the mycelium to dry and constricted. Growth of exposed fungus colonies was inhibited by VOCs, the inhibition rates of VOCs were 56.21% and 59.23%, 64.75% and 59.86%, 66.13% and 61.10%, 67.04% and 70.00%, respectively, against P. parasitica and A. alternata cultured in sealed plates for 2, 4, 6 and 8 d. When the zoospores of P. parasitica and ascospores of A. alternata exposed to these volatile components for 6 h, the germination was delayed and the growth was slow. The number of sporocyst produced by P. parasitica obviously reduced. Most conidiophores of A. alternata expanded abnormally as cystic structure, indicating the fungicidal nature of the volatiles. Moreover, VOCs could significantly inhibit the disease severities of tobacco black shank and brown spot on leaves tests. Direct fumigation for 40 h and 80 h, black shank disease incidence was 92.50% on control and 70.83% on leaves treated by VOCs, the inhibitory of spot expansion was 62.35%. Brown spot disease incidence was 88.33% on control and 60.80% on leaves treated by VOCs, lesions expanded slowly and inhibitory rate was 65.75%. SPME GC-MS analysis showed that seven components of the volatiles were identified, all of which are sesquiterpenes with C15H24 structure. They are dihydrocurcumene (CAS NO. 1461-02-5), (E)-β-famesene (CAS NO. 18794-84-8), γ-curcumene (CAS NO. 451-55-8), α-zingiberene (CAS NO. 495-60-3), π-bisabolene (CAS NO. 495-61-4), β-sesquiphellandrene (CAS NO. 20307-83-9) and γ-E-bisabolene (CAS NO. 53585-13-0). When AR03 was cultured for 1 d, the relative content of β-sesquiphellandrene was the highest (80.64%), followed by (E)-β-famesene and α-zingiberene, the relative content was 7.20% and 6.67%, respectively. With the extension of culturing time, the species of each component were the same, but the relative content was different. Except for dihydrocurcumene, the content of other components showed a decreasing trend, when cultured for 6 d, other ingredients decreased more than 50%, besides dihydrocurcumene keeping relatively stable. 【Conclusion】 VOCs produced by B. pumilus AR03 could develop an additive antifungal effect against fungal pathogens on tobacco. B. pumilus AR03 has potential as an important microbial resource for developing antifungal metabolites and new drugs.
DOI:10.1080/09583157.2014.960809URL [本文引用: 1]
[本文引用: 1]
DOI:10.1016/S0168-9452(03)00330-3URL [本文引用: 1]
DOI:10.1007/s12275-019-8517-9URL [本文引用: 1]
DOI:10.1111/1574-6968.12088URL [本文引用: 1]

Streptomyces sp. TD-1 was identified as Streptomyces alboflavus based on its morphological characteristics, physiological properties, and 16S rDNA gene sequence analysis. The antifungal activity of the volatile-producing S.alboflavus TD-1 was investigated. Results showed that volatiles generated by S.alboflavus TD-1 inhibited storage fungi Fusarium moniliforme Sheldon, Aspergillus flavus, Aspergillus ochraceus, Aspergillus niger, and Penicillum citrinum in vitro. GC/MS analysis revealed that 27 kinds of volatile organic compounds were identified from the volatiles of S.alboflavus TD-1 mycelia, among which the most abundant compound was 2-methylisoborneol. Dimethyl disulfide was proved to have antifungal activity against F.moniliforme by fumigation in vitro.
DOI:10.1016/j.soilbio.2004.10.021URL [本文引用: 1]
