摘要/Abstract
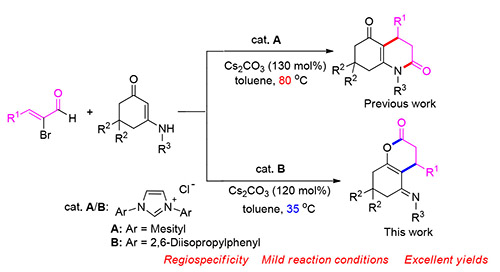
氮杂环卡宾(NHC)催化下α-溴代烯醛和烯胺酮的[3+3]环化反应的区域选择性受温度控制.在35℃时,该反应能够区域专一性地给出稠合吡喃酮衍生物,而不是文献报道的喹啉酮类化合物.该方法反应条件温和、底物范围广、产率高、操作简便,为吡喃酮骨架的高效构建提供了新思路.
关键词: 氮杂环卡宾(NHC), α-溴代烯醛, 烯胺酮, [3+3]环化
The regioselectivity of N-heterocyclic carbene (NHC)-catalyzed[3+3] annulation of α-bromoenal with enaminone is dependent on the temperature. The reactions performed at 35℃ could regiospecifically give the derivatives of fused pyranone instead of previously reported quinolones, which provided a new shortcut to pyranone with mild reaction condition, broad substrate scope, high yields and operational simplicity.
Key words: N-heterocyclic carbene (NHC), α-bromoenal, enaminone, [3+3]annulation
PDF全文下载地址:
点我下载PDF
