3.中国科学院生态环境研究中心,水污染控制实验室,北京 100085
1.School of Civil Engineering, University of South China, Hengyang 421001, China
2.State Key Joint Laboratory of Environment Simulation and Pollution Control, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, Beijing 100085, China
3.Department of Water Pollution Control, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, Beijing 100085, China
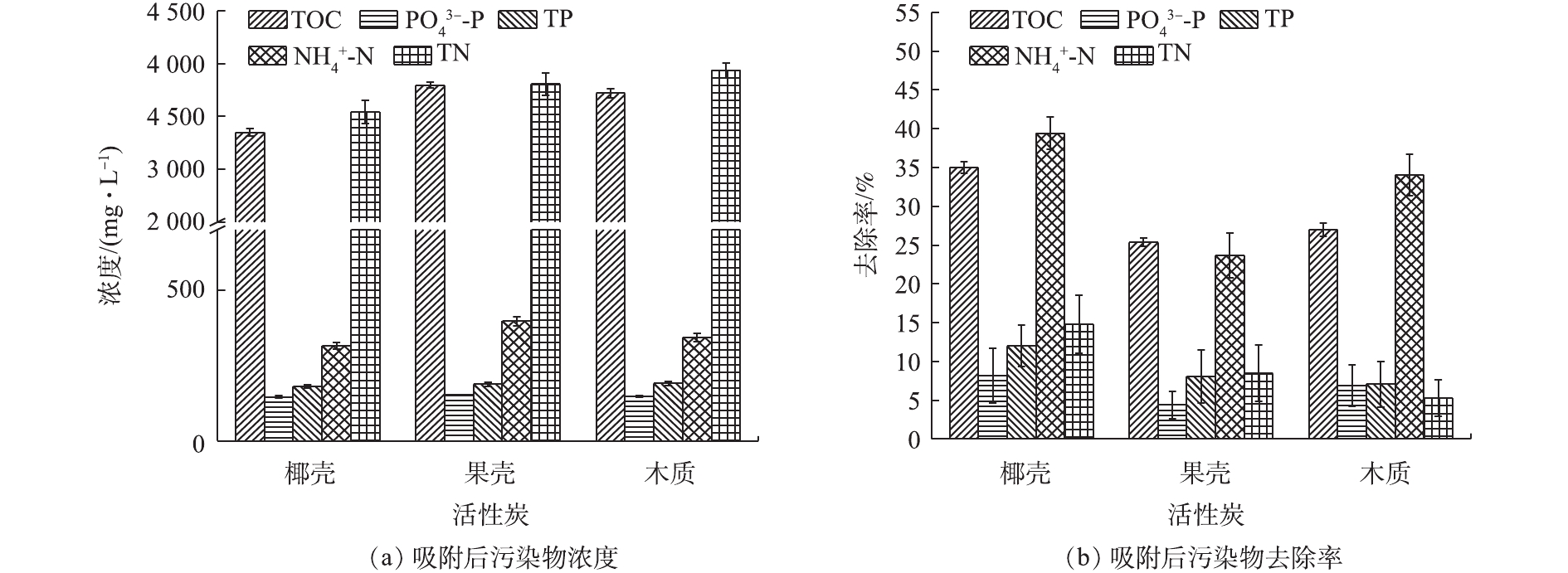
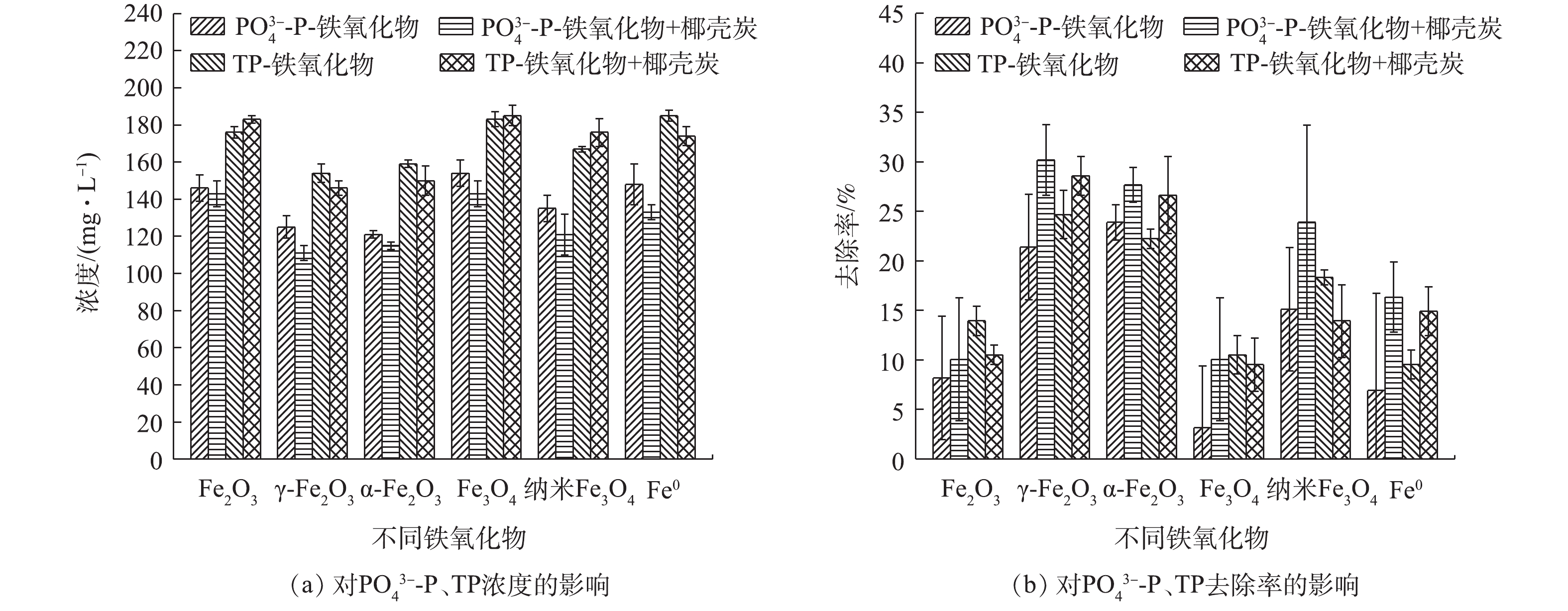
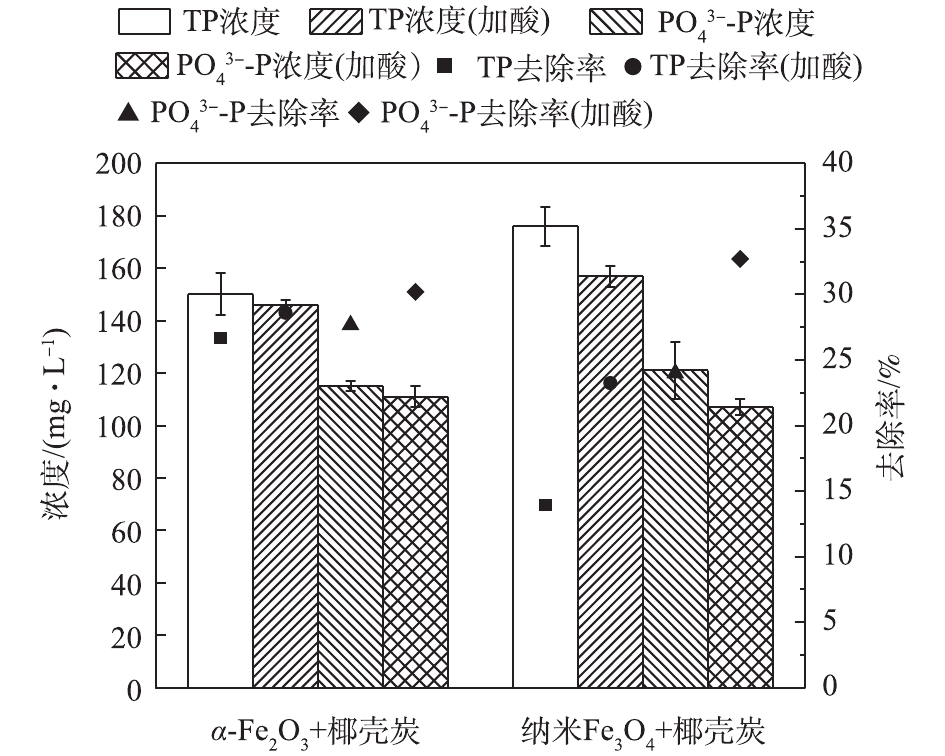
-N和TN的去除率可达35.02%、8.17%、11.98%、39.42%和14.79%;投加的铁氧化物对P的去除效果较好,去除率提高了14%,且酸预处理进一步提升了P的去除效果,去除率提高了9.29%;但在酸性条件下,有机物和
-N的吸附能力略有所下降;吸附过程对有机物的削减主要是由于活性炭的吸附及尿素的分解所致;N去除的主要机理是由于鸟粪石的沉淀所致;P削减的主要原因为铁氧化物表面的羟基位点吸附和鸟粪石沉淀,酸性条件可促进铁氧化物和
发生质子化过程,因此,其可进一步强化P的去除。综合上述结果,铁氧化物-活性炭吸附可有效去除尿液中有机物和P,是一种有效的尿液预处理工艺,以上结果可为尿液的处理和回收提供参考。
Three kinds of activated carbon (coconut shell, nutshell and wooden activated carbons) were applied for urine treatment, and the effect of iron oxides and urine acidification treatment on urine treatment by coconut shell activated carbons was carefully investigated in terms of N, P and organic matters. The result showed that coconut shell activated carbon had the best urine treatment performance due to its high specific surface area, and the removal rates of TOC,
-N and TN could reach 35.02%, 8.17%, 11.98%, 39.42% and 14.79%, respectively. The addition of iron oxides could lead to the increase of phosphorus removal rate by 14%, and the acidification pretreatment could further elevate TP removal rate by 9.29%. While under the acidic conditions, the adsorption ability of organics and
-N slightly decreased. The adsorption on activated carbon and urea decomposition were key reasons for TOC removal, and the struvite precipitation was the main reason for N removal, adsorption by the hydroxyl groups on the surface of iron oxide and struvite precipitation were the key reasons for P removal. Acidification pretreatment could promote the protonation between hydroxyl groups on iron oxide and phosphate and further enhance P removal. In conclusion, adsorption by iron oxides-activated carbon had excellent removal performances towards organic matters and P, which showed a great potential for urine pretreatment, it will provide reference for the treatment and recovery of urine.
.
Adsorption performance of different activated carbons toward urine
Effect of different iron oxides on TOC removal efficiency
Effect of urine pretreatment on TOC removal efficiency
| [1] | 董良飞, 蒋健钗, UDERT K M, 等. 尿液源头分离系统中的沉淀研究[J]. 环境工程学报, 2008, 2(7): 964-968. |
| [2] | 蒋善庆, 王晓昌, 李超, 等. 源分离尿液资源化利用与风险控制技术研究进展[J]. 安全与环境学报, 2014, 14(5): 174-182. |
| [3] | 朱国荣, 谢倍珍, 刘红. 载人深空探测活动中的尿液处理回收技术分析[J]. 深空探测学报, 2018, 5(6): 582-590. |
| [4] | 杨祺, 张文瑞, 于锟锟. 空间站尿液处理技术研究及进展[J]. 真空与低温, 2014, 20(6): 315-318. doi: 10.3969/j.issn.1006-7086.2014.06.002 |
| [5] | 王健行. 密闭生保系统中基于盐回收的纳滤膜技术及膜污染防治[R]. 北京: 中国科学院生态环境研究中心博士后出站报告, 2016. |
| [6] | SOLANKI A, BOYER T H. Physical-chemical interactions between pharmaceuticals and biochar in synthetic and real urine[J]. Chemosphere, 2019, 218: 818-826. doi: 10.1016/j.chemosphere.2018.11.179 |
| [7] | PRADHAN S K, MIKOLAA, CAHALA R. Nitrogen and phosphorus harvesting from human urine using a stripping, absorption, and precipitation process[J]. Environmental Science & Technology, 2017, 51(9): 5165-5171. |
| [8] | 温国期, 胡正义, 刘小宁, 等. 改性沸石吸附柱去除和回收脱磷尿液废水中氨氮试验研究[J]. 农业环境科学学报, 2013, 32(12): 2488-2494. doi: 10.11654/jaes.2013.12.023 |
| [9] | LAIRD D, FLEMING P, WANG B, et al. Biochar impact on nutrient leaching from a midwestern agricultural soil[J]. Geoderma, 2010, 158(3/4): 436-442. |
| [10] | XU K, LIN F, DOU X, et al. Recovery of ammonium and phosphate from urine as value-added fertilizer using wood waste biochar loaded with magnesium oxides[J]. Journal of Cleaner Production, 2018, 187: 205-214. doi: 10.1016/j.jclepro.2018.03.206 |
| [11] | KUSIOR A, MICHALEC K, JELEN P, et al. Shaped Fe2O3 nanoparticles-synthesis and enhanced photocatalytic degradation towards RhB[J]. Applied Surface Science, 2019, 476: 342-352. doi: 10.1016/j.apsusc.2018.12.113 |
| [12] | 吴承慧, 陈长安, 高旭波, 等. 改性纳米级Fe3O4对地下水中氟的吸附性能研究[J]. 环境科学与技术, 2019, 42(6): 82-88. |
| [13] | 张斌, 韩宇霞, 赵斯琴, 等. 纳米γ-氧化铁的制备及其吸附性能研究[J]. 无机盐工业, 2019, 51(6): 76-79. |
| [14] | 黎想, 任彦瑛, 丁琳洁. 预磁化零价铁活化过硫酸盐体系降解双氯芬酸钠[J]. 环境工程学报, 2019, 13(12): 2808-2815. doi: 10.12030/j.cjee.201901116 |
| [15] | 甘莉, 曹丹, 金晓英, 等. 离子型表面活性剂改性绿色合成单分散纳米氧化铁的制备及其吸附磷的性能[J]. 环境科学学报, 2015, 35(8): 2442-2449. |
| [16] | 李婷, 朱易春, 康旭, 等. 海绵铁还原微污染源水中硝酸盐氮的影响因素研究[J]. 工业水处理, 2016, 36(11): 85-89. |
| [17] | JIA Z, LIU J, WANG Q, et al. Synthesis of 3D hierarchical porous iron oxides for adsorption of congo red from dye wastewater[J]. Journal of Alloys and Compounds, 2015, 622: 587-595. doi: 10.1016/j.jallcom.2014.10.125 |
| [18] | WANG J, XU L, ZHANG Z, et al. Shape-controlled synthesis of α-Fe2O3 nanocrystals for efficient adsorptive removal of Congo red[J]. RSC Advances, 2015, 5(61): 49696-49702. doi: 10.1039/C5RA06324H |
| [19] | 刘锦华. 四种吸附材料对蓝/红物质的吸附作用及黑臭水体光学特性研究[D]. 广州: 广东工业大学, 2018. |
| [20] | UDERT K M, LARSEN T A, BIEBOW M, et al. Urea hydrolysis and precipitation dynamics in a urine-collecting system[J]. Water Research, 2003, 37(11): 2571-2582. doi: 10.1016/S0043-1354(03)00065-4 |
| [21] | LIU Z, ZHAO Q, WANG K, et al. Urea hydrolysis and recovery of nitrogen and phosphorous as MAP from stale human urine[J]. Journal of Environmental Sciences, 2008, 20(8): 1018-1024. doi: 10.1016/S1001-0742(08)62202-0 |
| [22] | 曾俊钦, 邱春生, 孙力平, 等. 储存控制条件对尿液氮磷的影响[J]. 环境工程学报, 2016, 10(10): 5605-5610. doi: 10.12030/j.cjee.201505094 |
| [23] | ZHU K, FU H, ZHANG J, et al. Studies on removal of ${{\rm{NH}}_4^{+} } $  -N from aqueous solution by using the activated carbons derived from rice husk[J]. Biomass and Bioenergy, 2012, 43: 18-25. doi: 10.1016/j.biombioe.2012.04.005 -N from aqueous solution by using the activated carbons derived from rice husk[J]. Biomass and Bioenergy, 2012, 43: 18-25. doi: 10.1016/j.biombioe.2012.04.005 |
| [24] | ZHANG S, ZHAO X, NIU H, et al. Superparamagnetic Fe3O4 nanoparticles as catalysts for the catalytic oxidation of phenolic and aniline compounds[J]. Journal of Hazardous Materials, 2009, 167(1): 560-566. |
| [25] | 宋冬. 零价铁强化低碳源城市污水处理厂脱氮除磷效果研究[D]. 西安: 长安大学, 2016. |
| [26] | 付丰连. 零价铁处理污水的最新研究进展[J]. 工业水处理, 2010, 30(6): 1-4. doi: 10.3969/j.issn.1005-829X.2010.06.001 |
| [27] | HUANG X. Intersection of isotherms for phosphate adsorption on hematite[J]. Journal of Colloid and Interface Science, 2004, 271(2): 296-307. doi: 10.1016/j.jcis.2003.12.007 |
| [28] | ZHU Z, HUANG C P, ZHU Y, et al. A hierarchical porous adsorbent of nano-α-Fe2O3/Fe3O4 on bamboo biochar (HPA-Fe/C-B) for the removal of phosphate from water[J]. Journal of Water Process Engineering, 2018, 25: 96-104. doi: 10.1016/j.jwpe.2018.05.010 |
| [29] | 席宏波, 廖娣劼, 尚海涛, 等. 纳米铁除磷的影响因素及吸附模式研究[J]. 给水排水, 2008, 44(S1): 191-195. |
| [30] | 张颖纯, 王伟. 纳米零价铁颗粒除磷反应机理[J]. 环境工程学报, 2015, 9(5): 2041-2047. doi: 10.12030/j.cjee.20150502 |
| [31] | 冯皓迪, 丁一珊, 崔梦萦, 等. MAP法沉淀回收尿液中氮磷的研究[J]. 环境工程, 2016, 34(S1): 162-166. |
| [32] | 李春霞, 赵成坚, 祝迎春. 基于K2Cr2O7/H2SO4体系的尿预处理剂对尿液的影响[J]. 航天医学与医学工程, 2016, 29(2): 133-136. |
| [33] | 刘斌, 顾洁, 邱盼, 等. 稻壳与脱硅稻壳活性炭特性及对有机物吸附[J]. 林产化学与工业, 2014, 34(5): 27-34. |
| [34] | LIANG H, LIU K, NI Y. Synthesis of mesoporous α-Fe2O3 using cellulose nanocrystals as template and its use for the removal of phosphate from wastewater[J]. Journal of the Taiwan Institute of Chemical Engineers, 2017, 71: 474-479. doi: 10.1016/j.jtice.2016.12.008 |
| [35] | LI G, GAO S, ZHANG G, et al. Enhanced adsorption of phosphate from aqueous solution by nanostructured iron(III)-copper(II) binary oxides[J]. Chemical Engineering Journal, 2014, 235: 124-131. doi: 10.1016/j.cej.2013.09.021 |
| [36] | 蒋善庆, 王晓昌. 黄土颗粒吸附水解尿液中磷酸盐特性及机制[J]. 化工进展, 2017, 36(7): 2667-2675. |








 下载:
下载: 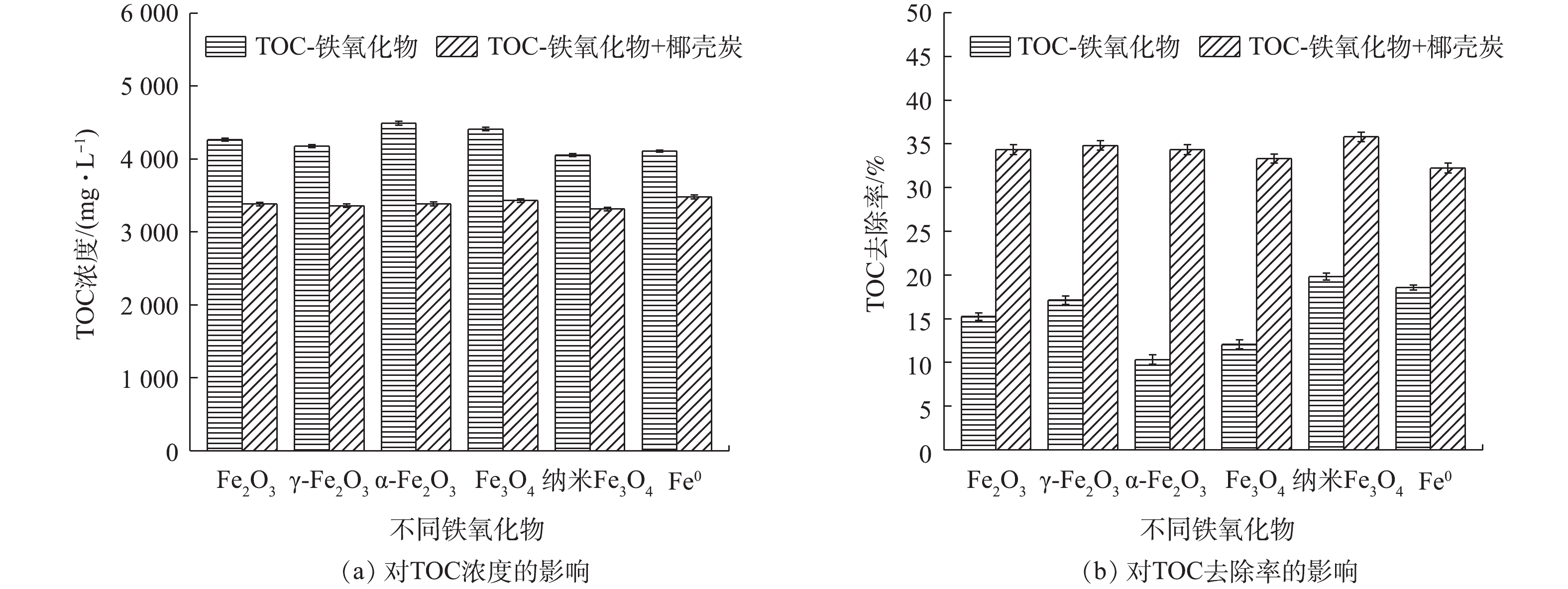




 点击查看大图
点击查看大图