全文HTML
--> --> -->在全固态电池中, 固态电解质作为重要组成部分, 是影响全固态电池性能的关键因素. 近几年, 关于锂/钠固态电解质的报道层出不穷, 经典的固态电解质体系主要集中在氧化物和硫化物[11-23]. 其中, 高离子电导率的固态电解质以硫化物为主[12,13,15,18,20], 氧化物通常需要1000 ℃以上热处理来提升离子电导率[16,24]. 反钙钛矿型Li3OCl和Li3OCl1–xBrx是一类具有超离子电导率的固态电解质, 室温离子电导率在10–3 S·cm–1以上[25]. 除此之外, Li3OCl1–xBrx也展现出良好的电化学稳定性和低熔点(约280 ℃), 使其在较低温度下处理即可减小晶界阻抗[25].Li3OCl1–xBrx的优异特性激起了研究者对富钠反钙钛矿材料的研究兴趣, 一系列富钠反钙钛矿Na3–2xMxOA (M = Ca, Sr; A = Cl, Br, I), Na3SO4F等已经被成功合成[26,27]. 化学调控和放电等离子体烧结(spark plasma sintering, SPS)等可用于提升材料离子电导率[26-29]. 尽管优化后, Na3OBrxI1–x的离子电导率在200 ℃可达1.89 × 10–3 S·cm–1, 但是室温下仍然低至10–7—10–6 S·cm–1[26]. Braga等[30,31]通过在前驱体中引入少量去离子水, 制备玻璃态电解质A2.99M0.005OCl1–x(OH)x(A = Li或Na, M = Ba或Ca), 离子电导率在室温下超过10–2 S·cm–1, 但是该材料属于完全不定形态, 结构信息难以确定. 近期, 基于团簇阴离子




本文在较低合成温度(250 ℃)成功制备富钠反钙钛矿Na3OBrxI1–x (0 < x < 1), 且材料离子电导率与文献相比有较大提升, 经热处理之后, Na3OBr0.3I0.7在100 ℃的离子电导率为1.47 × 10–3 S·cm–1. 然而, 随着温度降低, Na3OBr0.3I0.7的离子电导率出现跳变, 固态核磁(NMR)分析表明, 该现象可能与材料复杂的结构对称性和钠位变化有关. 进一步对Na3OBr0.5I0.5进行电化学性能研究, 结果显示, Na3OBr0.5I0.5材料具有良好的钠金属相容性, 且能保证固态电池稳定运行. 通过离子液体进行界面修饰后, Na-Na对称电池在0.5 A·cm–2电流密度下稳定循环, 过电位在80 mV以下; TiS2/Na3OBr0.5I0.5/Na-Sn全电池在50 ℃, 0.1 C(1 C = 240 mA/g)条件下, 首次放电比容量为190 mAh·g–1, 循环10圈后, 比容量保持在127 mAh·g–1左右. 稳定的电化学性能表明富钠反钙钛矿材料在固态钠电池方面拥有较好的应用前景, 对固态钠电池的发展具有重要意义.
2.1.材料合成
富钠反钙钛矿固态电解质Na3OBrxI1–x以Na2O (Alfa Aesar; AR, 80% Na2O, 20%Na2O2), NaBr (General-Reagent; AR, >99% purity)和NaI (General-Reagent; AR, >99% purity)为前驱体合成, 制备过程参照Hipplern等[41]的报道, 但合成温度大幅降低, 更利于实际生产. 具体过程如下: 在手套箱内(H2O, O2 < 0.1 ppm)将Na2O, NaBr, NaI按照一定摩尔比称量后用研钵研磨均匀, 然后将混合粉末转移至氧化锆内衬的球磨罐中, 在350 rpm转速下混合10 h, 将混合好的粉末压成片后密封在石英管中, 在250 ℃下煅烧12 h, 自然冷却后即得到所需样品. 按计量比不同将材料标记为Na3OBr0.7I0.3, Na3OBr0.5I0.5, Na3OBr0.3I0.7.2
2.2.表征
X射线衍射(X-ray diffraction, XRD)用于材料晶体结构分析, 2θ范围为25°—70°, 样品制备过程中用有机薄膜覆盖, 用于隔绝空气; 扫描电子显微镜(scanning electron microscope, SEM)用于观察电解质片冷压和热压之后的微观形貌; 固态核磁共振(nuclear magnetic resonance, NMR)用于分析不同温度下材料结构变化.2
2.3.电化学测试
离子电导率测试过程中, 电解质片的制备如下: 每个样品取70 mg左右置于直径10 mm的聚醚醚酮(poly-ether-ether-ketone, PEEK)圆柱模具中, 在300 MPa压力下冷压成片, 将成型的样品与不锈钢杆集流体接触, 密封在金属模具中, 并在100 ℃真空烘箱中保持12 h, 然后用输力强1260 A测试样品在不同温度下的交流(AC)阻抗谱, 测试频率范围为0.1 Hz—10 MHz. 组装Na/Na3OBr0.5I0.5/Na对称电池用于评估电解质与钠金属之间的电化学稳定性, 电解质与钠金属之间引入6 μL/cm2离子液体(ionic liquid, IL)润湿界面, 其中离子液体成分为0.8 M NaTFSI溶于PP13FSI. 对称电池分别在0.05和0.5 mA/cm2电流密度下进行钠沉积测试.2
2.4.固态钠电池的组装
以TiS2、科琴黑、聚(偏二氟乙烯)(poly(vinylidene fluoride), PVDF)分别为活性物质、导电剂、粘结剂, 质量比为8∶1∶1, 以N-甲基吡咯烷酮(N-Methyl pyrrolidone, NMP)为溶剂混合均匀后形成复合正极浆料, 将浆料涂覆在涂碳铝箔上, 70 ℃干燥12 h得到TiS2复合正极片, 其中活性物质含量约为1 mg·cm–2. Na-Sn合金负极通过将Na箔和Sn箔热压后制得, 具体为: 将Na箔和Sn箔堆叠之后置于热压机上, 在80 ℃, 15 MPa条件下保持15 min即得到Na-Sn合金. 固态电池组装过程在手套箱中进行, 首先, 将Na3OBr0.5I0.5以2.3节中的方式冷压成片, 然后将正负极分别置于电解质片的两侧, 并提供38 MPa左右压力以确保材料接触, 最后在正负极两侧分别添加6.4 μL·cm–2离子液体. 电池恒流充放电测试在0.1 C倍率, 50 ℃条件下进行, 测试设备为新威尔充放电测试通道(CT4008), 所有电池测试过程均在手套箱中进行.3.1.XRD表征
如图1所示, 合成的Na3OBrxI1–x (x = 0.3, 0.5, 0.7)样品的XRD图谱与文献报道的反钙钛矿结构一致[26], 通过简单的合成方法得到的样品为纯相Na3OBrxI1–x, 不含其他杂质. 随着结构中I含量增加, 衍射峰位置向左偏移, 根据布拉格方程2dsinθ = nλ(d为晶面间距; θ为入射线、反射线与反射晶面之间的夹角; λ为波长; n为反射级数), 衍射峰左移, 说明θ减小, 即可判断d增大, 得出晶格尺寸增大的结论, 而晶格尺寸增大是因为I–的半径比Br–大, 说明I成功掺杂. 另一方面, 本文中材料XRD图谱没有观察到衍射峰劈裂, 而23Na NMR谱却显示有两个Na信号(图3(a)), 因此很难将合成的Na3OBrxI1–x归结为立方相或文献报道的P2/m单斜相空间群[26]. Na3OBrxI1–x材料结构将在之后结合23Na NMR结果进一步讨论.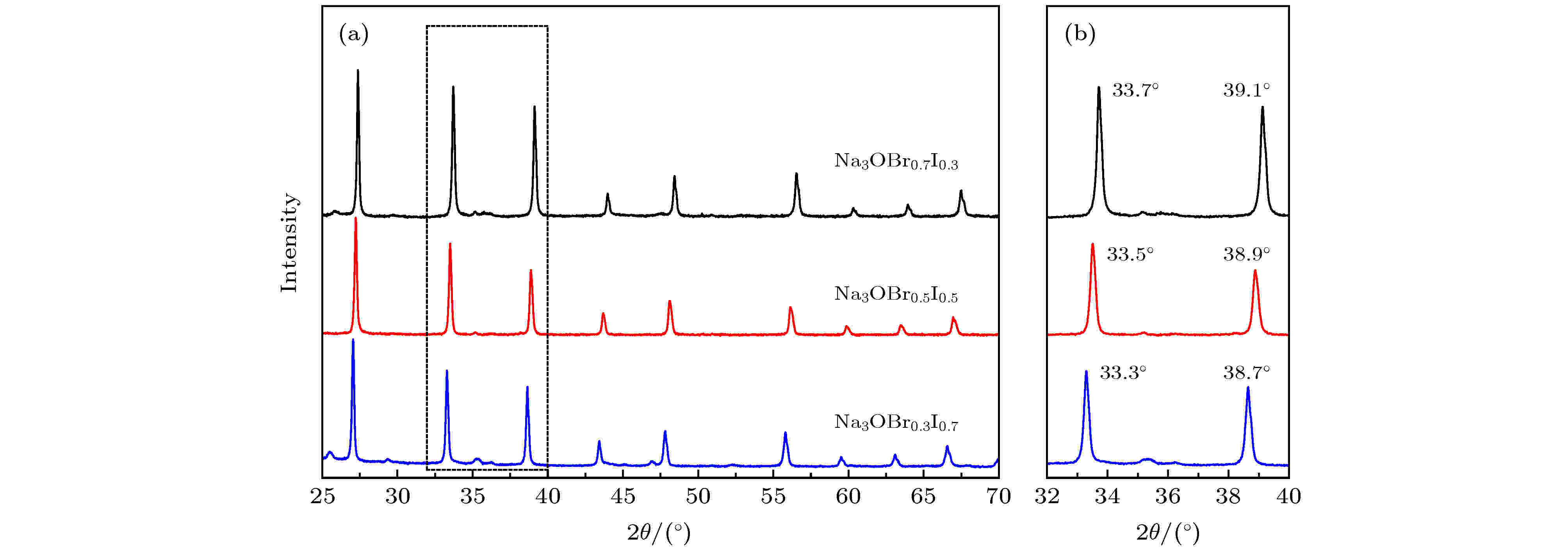 图 1 (a)合成的反钙钛矿Na3OBrxI1–x (x = 0.3, 0.5, 0.7)样品的XRD图谱; (b)图(a)的局部放大图
图 1 (a)合成的反钙钛矿Na3OBrxI1–x (x = 0.3, 0.5, 0.7)样品的XRD图谱; (b)图(a)的局部放大图Figure1. (a) The X-ray diffraction (XRD) patterns of synthezied anti-perovskites Na3OBrxI1–x (x = 0.3, 0.5, 0.7); (b) local zoom of Fig. (a).
2
3.2.钠离子电导率
研究表明样品的处理方式对离子电导率的测试至关重要, 本文中Na3OBrxI1–x的离子电导率通过对材料在100 ℃热压之后, 利用交流阻抗法测得. Na3OBr0.5I0.5冷压和热压片的致密度对比如表1所列, 通过计算电解质片的实际质量和体积, 得出冷压和热压电解质片的密度分别为2.11和2.55 g/cm3, 致密度分别为69%和83%, 这表明热压能促进样品的致密化. 如图2(a)和图2(b)所示, 通过SEM对Na3OBr0.5I0.5电解质冷压和热压片进行微观结构观察, 可以看出, 热压之后的电解质片具有更平整致密的表面结构.| 密度/ g·cm–3 | 致密度 | |
| 冷压 | 2.11 | 69% |
| 热压 | 2.55 | 83% |
| 真实密度* | 3.06 | — |
| *真实密度基于XRD谱得到的晶格参数计算, 晶格参数计算基于简单的立方相[26], 忽略结构对称性破坏引起的细微变化. | ||
表1Na3OBr0.5I0.5在冷压和热压下的密度
Table1.Density of hot- and cold-pressed Na3OBr0.5I0.5
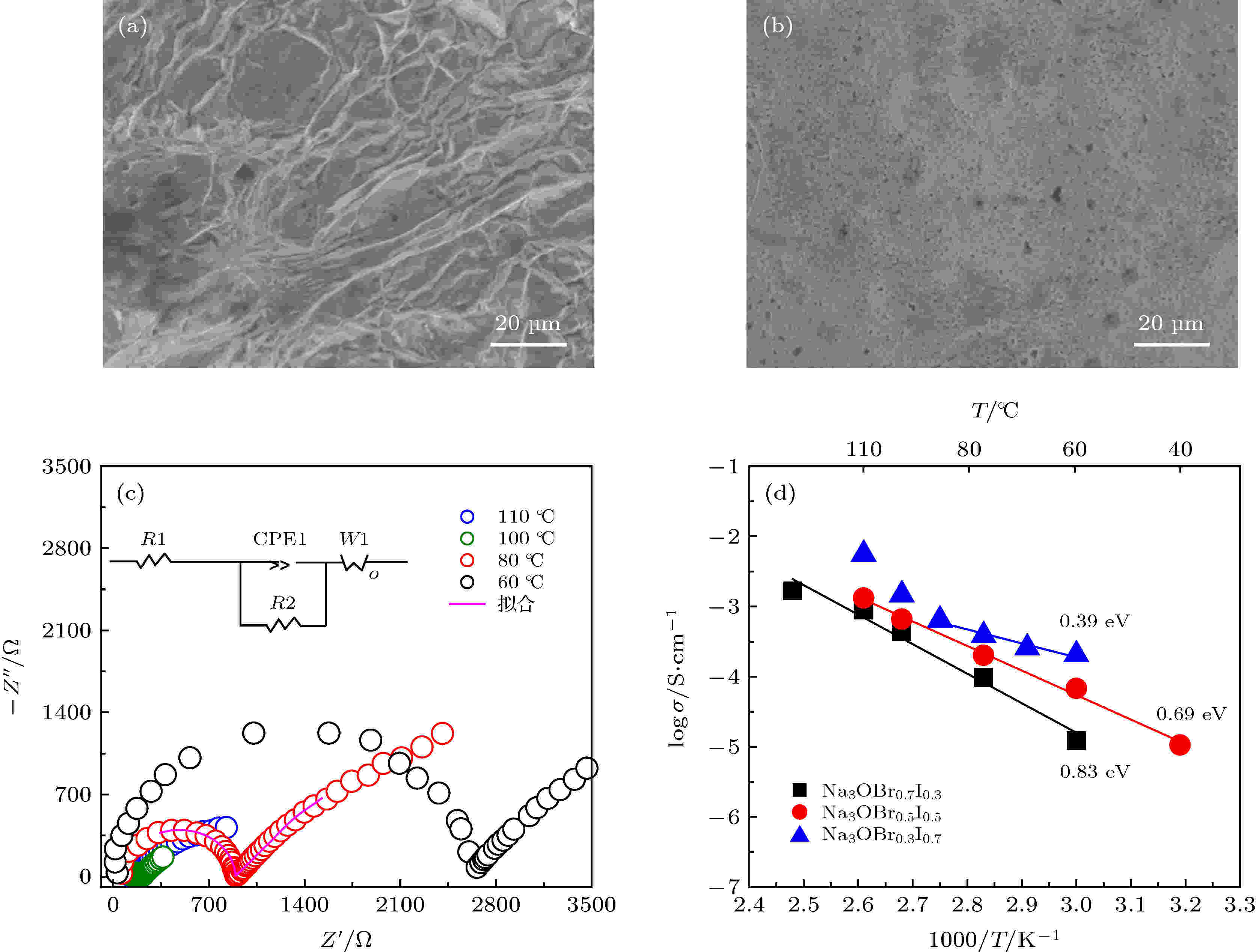 图 2 通过(a)冷压和(b)热压方法制备得到的Na3OBr0.5I0.5电解质片的SEM图; (c)不同温度下测得的热压Na3OBr0.5I0.5片的Nyquist曲线; (d) Na3OBrxI1–x (x = 0.3, 0.5, 0.7)的logσ与1000/T对应曲线
图 2 通过(a)冷压和(b)热压方法制备得到的Na3OBr0.5I0.5电解质片的SEM图; (c)不同温度下测得的热压Na3OBr0.5I0.5片的Nyquist曲线; (d) Na3OBrxI1–x (x = 0.3, 0.5, 0.7)的logσ与1000/T对应曲线Figure2. SEM images of (a) cold-pressed and (b) hot-pressed Na3OBr0.5I0.5 solid electrolyte pellets; (c) Nyquist plots of hot-pressed Na3OBr0.5I0.5 measured at different temperatures; (d) logσ versus 1000/T plots for Na3OBrxI1–x (x = 0.3, 0.5, 0.7).
交流阻抗用于测试热压电解质片在不同温度下的离子电导率, 计算公式如下:
L-电解质片厚度, 单位: cm;
S-电解质片面积, 单位: cm2;
R-Nyquist曲线中阻抗值, 单位: Ω.
图2(c)为Na3OBr0.5I0.5在不同温度下的Nyquist曲线, 样品阻抗随着温度升高逐渐减小, 各材料离子电导率具体数值如表2所列. 图2(d)反应了各材料的logσ与1000/T对应关系曲线, 并计算各电解质活化能. 可以看出, 随着I含量增加, 材料离子电导率呈上升趋势, 活化能也逐渐减小, 这可能归因于I的引入使材料晶格尺寸增加, 更利于Na+离子传输; 但是, 当I含量增加至0.7时, 材料的离子电导率在温度变化过程中更容易发生跳变. 下面将进一步探讨不同温度下材料的结构变化规律.
| 温度/℃ | 离子电导率/ S·cm–1 | ||
| Na3OBr0.7I0.3 | Na3OBr0.5I0.5 | Na3OBr0.3I0.7 | |
| 130 | 1.67 × 10–3 | — | — |
| 110 | 8.96 × 10–4 | 1.32 × 10–3 | 5.55 × 10–3 |
| 100 | 4.50 × 10–4 | 6.56 × 10–4 | 1.47 × 10–3 |
| 80 | 9.73 × 10–5 | 2.01 × 10–4 | 3.93 × 10–4 |
| 60 | 1.22 × 10–5 | 6.78 × 10–5 | 2.05 × 10–4 |
| 40 | — | 1.06 × 10–5 | — |
表2Na3OBrxI1–x (x = 0.3, 0.5, 0.7)离子电导率
Table2.Ionic conductivity of Na3OBrxI1–x (x = 0.3, 0.5, 0.7).
2
3.3.不同温度相表征
固态NMR和原位高温XRD用于研究材料变温过程中离子电导率的跳变现象. 由于离子电导率测试过程中, Na3OBr0.3I0.7样品的跳变现象最为明显, 因此选择该样品作为研究对象. 如图3(b)所示, 温度从25 ℃上升至100 ℃的过程中, 材料的XRD图谱保持一致, 没有观察到与相转变有关的变化. 同时, 尽管Na3OBr0.5I0.5被归结为单斜相(P2/m空间群)[26], 但是Na3OBr0.3I0.7样品的XRD图谱中没有观察到立方相对称性破坏引起的衍射峰劈裂, 因此推测Na3OBr0.3I0.7为立方相反钙钛矿结构. 与XRD结果相反, 图3(a)NMR谱中23Na信号显示, 在Na3OBr0.3I0.7中至少有两个不同的Na位, 与Na3OBr的一致[32], 表明Na3OBr和Na3OBr0.3I0.7的低对称结构. 当温度升高时, 10 — 12 ppm处信号的相对强度减弱, 并向低δ方向偏移, 这暗示了与Na扩散对应的Na位变化. 另外, 基于XRD精修, Na3OBr和Na3OBr0.5I0.5分别归属为只有一个Na位的立方相反钙钛矿结构和含有三个Na位的单斜相空间群[26]. 这些结果显示Na3OBrxI1–x材料结构和对称性的复杂性, 需要更多实验和研究去确认Na3OBrxI1–x材料精确的晶体结构, 这将对深入理解离子电导率随温度变化的机理, 以及提升离子电导率, 尤其是室温离子电导率方面具有重要意义.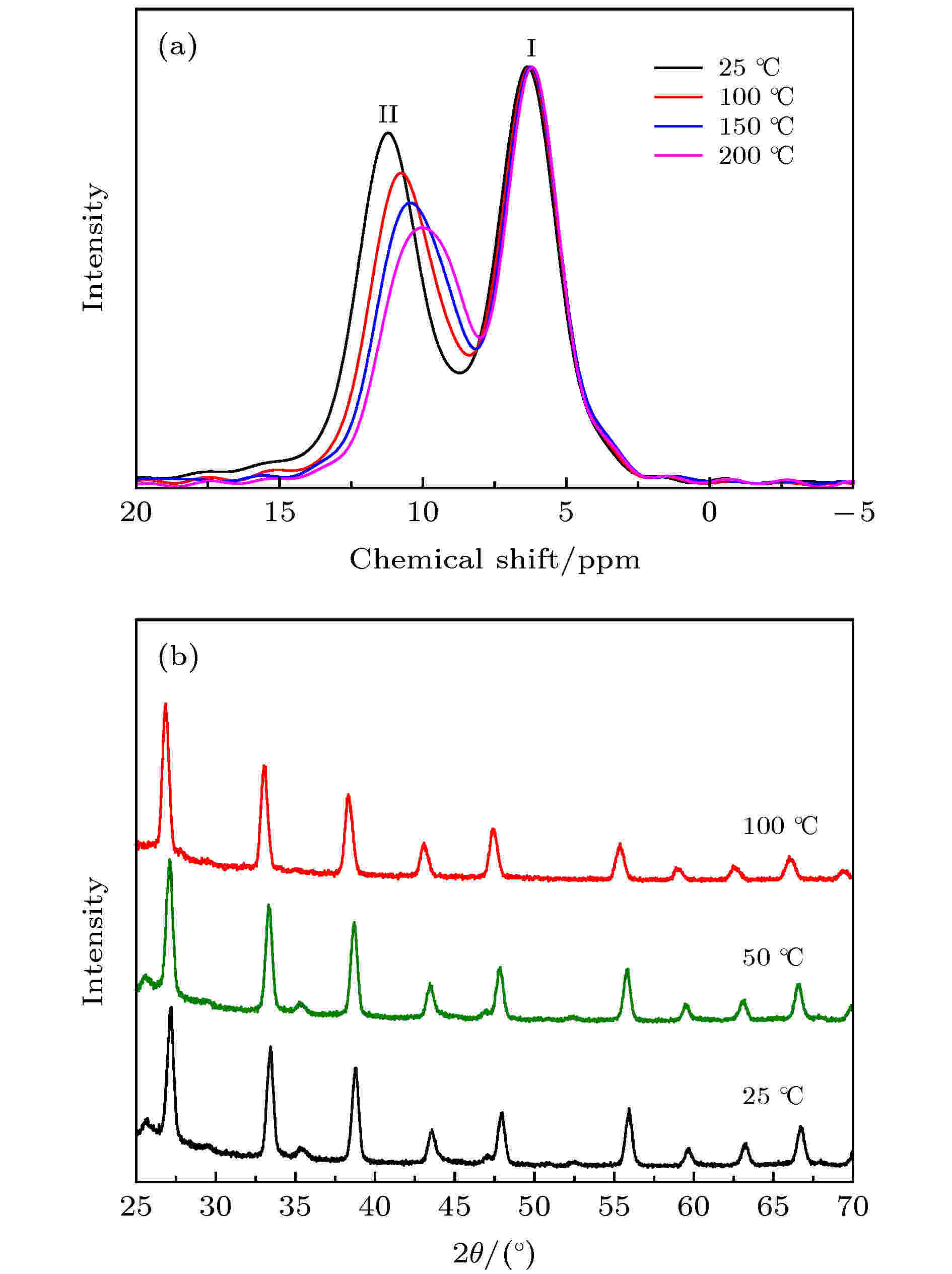 图 3 Na3OBr0.3I0.7在不同温度下的 (a)固态核磁图谱; (b)XRD图谱
图 3 Na3OBr0.3I0.7在不同温度下的 (a)固态核磁图谱; (b)XRD图谱Figure3. (a) Solid state 23Na NMR spectra and (b) XRD patterns of Na3OBr0.3I0.7 at different temperature.
2
3.4.电化学性能
固态电解质的最终目的是应用到固态电池中, 目前, 将富钠反钙钛矿钠离子固态电解质应用到固态电池中的报道较少. Na3OBr1–xIx的离子电导率显示其具有在中温固态钠电池中应用的潜能(40—80 ℃离子电导率接近或高于10–4 S·cm–1), 因此, 通过组装Na-Na对称电池和TiS2全电池, 在50 ℃条件下进行测试, 评估其对钠金属相容性以及在固态钠电池中的性能.x = 0.3时, 样品离子电导率偏低; x = 0.7时, 材料离子电导率随温度变化更易发生跳变. 综合考虑, 选择Na3OBr0.5I0.5作为固态电解质组装固态电池. 由于Na3OBr0.5I0.5亲钠性较差, Na/Na3OBr0.5I0.5/Na对称电池的阻抗非常大, 如图4(a)所示. 这为电化学性能测试和钠金属负极的使用带来了挑战, 而界面工程是解决这一问题的有效方法. 本工作的主要目的是评估Na3OBr0.5I0.5作为固态电解质的电化学性能, 因此引入少量离子液体修饰电解质/电极界面, 减小电池阻抗. 如图4(b)所示, 添加少量离子液体后, 对称电池阻抗显著降低. 如图4(c)所示, 在Na沉积/剥离过程中, 对称电池展现出稳定的过电位曲线, 且当电流密度从0.05 mA·cm–2增加到0.5 mA·cm–2时, 电池仍然能稳定运行, 不发生短路现象, 表明电解质对钠金属的电化学稳定性较好. 将Na3OBr0.5I0.5作为固态电解质组装在分别以TiS2和Na-Sn合金作为正负极的固态电池中, 同样引入少量离子液体润湿电解质/电极界面, 电池在50 ℃, 0.1 C倍率下运行. 添加离子液体[42]及在50 ℃条件运行是为了减小界面和电解质层阻抗. 如图4(d)所示, 固态钠电池初始放电比容量为190 mAh·g–1, 非常接近TiS2的理论比容量, 循环10圈后, 放电比容量保持在127 mAh·g–1. 上述结果显示, Na3OBr0.5I0.5拥有良好的电化学性能, 证明其具有在固态钠电池中应用的潜力. 电池容量衰减可能源于固态电解质与电极材料间较差的界面接触, 在该电池系统中, 电极材料在电池运行过程中会产生较大的体积变化, 且氧化物电解质硬度较大的特点不利于构建稳定的接触界面. 尽管低熔点能使固态电解质离子电导率提升变得更简单, 但是氧化物特性使其难以构筑较低阻抗的固态电解质/活性材料界面, 关于这方面的研究仍将继续.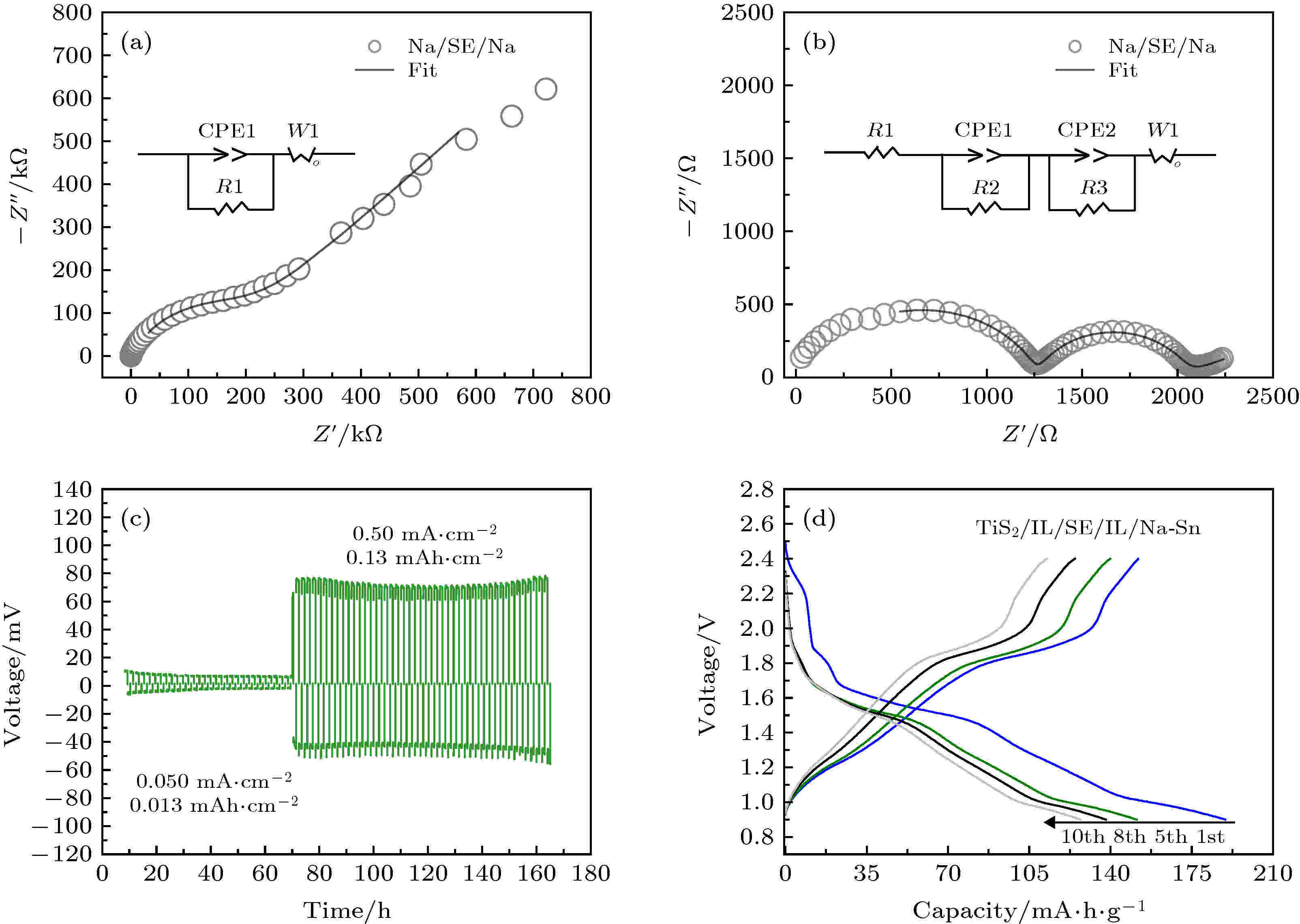 图 4 (a) Na/Na3OBr0.5I0.5/Na对称电池的电化学阻抗谱; (b) 添加了离子液体的Na/IL/Na3OBr0.5I0.5/IL/Na对称电池的电化学阻抗谱; (c) Na/IL/Na3OBr0.5I0.5/IL/Na对称电池在不同电流密度下的充放电曲线; (d) TiS2/IL/Na3OBr0.5I0.5/IL/Na-Sn在50 ℃, 0.1 C条件下充放电曲线
图 4 (a) Na/Na3OBr0.5I0.5/Na对称电池的电化学阻抗谱; (b) 添加了离子液体的Na/IL/Na3OBr0.5I0.5/IL/Na对称电池的电化学阻抗谱; (c) Na/IL/Na3OBr0.5I0.5/IL/Na对称电池在不同电流密度下的充放电曲线; (d) TiS2/IL/Na3OBr0.5I0.5/IL/Na-Sn在50 ℃, 0.1 C条件下充放电曲线Figure4. (a) Electrochemical impedance plot of Na/Na3OBr0.5I0.5/Na symmetrical cell; (b) electrochemical impedance plot of Na/IL/Na3OBr0.5I0.5/IL/Na symmetrical cell with ionic liquid; (c) charge-discharge curves of Na/IL/Na3OBr0.5I0.5/IL/Na symmetrical cell at different current density; (d) charge-discharge curves of TiS2/IL/Na3OBr0.5I0.5/IL/Na-Sn operated at 50°C, 0.1 C.
