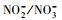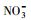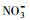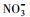, 张蓬2,3, 葛林科2,3
, 张蓬2,3, 葛林科2,3
 , 董倩倩1,3, 王德高1
, 董倩倩1,3, 王德高1

1. 大连海事大学环境科学与工程学院, 大连 116026;
2. 陕西科技大学环境科学与工程学院, 西安 710021;
3. 国家海洋环境监测中心, 国家环境保护近岸海域生态环境重点实验室, 大连 116023
收稿日期: 2019-11-10; 修回日期: 2020-01-08; 录用日期: 2020-01-08
基金项目: 国家自然科学基金(No.21976045,21577029,41476084);国家水体污染控制与治理科技重大专项(No.2017ZX07602-001)
作者简介: 张爽(1994-), 女, E-mail:18804203150@163.com
通讯作者(责任作者): 葛林科(1980—), 男, 博士, 教授, 主要研究方向为新型有机污染物环境光化学行为.主持国家自然科学基金项目3项, 发表论文20余篇.E-mail:gelinke@sust.edu.cn
王德高(1977—), 男, 教授, 主要研究方向为新型有毒物质的污染监测和环境行为.主持完成国家自然科学基金两项, 发表SCI期刊收录论文50篇, 出版学术专著1部.E-mail:degaowang@dlmu.edu.cn
摘要:冰雪是一类重要且普遍的环境介质,而冰雪环境光化学是一门新兴的学科.在相同光照条件下比较冰中和水中有机污染物的光化学行为,有助于揭示冰雪光化学与水环境光化学之间的异同.本文以四环素(TC)为模型化合物,比较了模拟日光(λ>290 nm)照射下不同水体冰相和水相中TC的光降解动力学,研究了不同相中溶解性物质(腐殖酸(HA)、Cl-、NO3-和Fe(III))对光降解的影响及作用机制,以揭示冰雪环境光化学和水环境光化学的异同.结果表明,冰中和水中TC光解遵循准一级反应动力学,纯水冰中表观光解量子产率为4.76×10-3,高于纯水中表观光解量子产率(3.85×10-3).在不同水体中,冰相TC的光解快慢顺序为海水冰中>淡水冰中>纯水冰中,而在水相中光解快慢顺序与冰相不同,为淡水中>海水中>纯水中.通过考察主要溶解性物质对冰/水中TC光降解动力学的影响发现,无论是在冰相还是水相,Cl-、HA、NO3-和Fe(III)均加快了TC的光降解,且促进作用随浓度的升高而增强.但在水相与冰相中每个因素促进的程度却存在差异,相对于水环境,冰中HA、NO3-、Fe(III)敏化作用较大,对TC光降解的促进作用较强;而冰中Cl-对TC光解的促进作用较水相中弱.这些溶解性物质对TC光解的促进作用能够解释海水冰、淡水冰与海水、淡水中TC相对于纯水冰相/水相具有较强的光降解能力.进一步地,将实验数据外推到实际环境,在35°~50°N地区的仲冬季,冰雪表面和水体表层TC光化学降解的半减期分别为15.4~38.9 min和19.0~48.0 min,其不仅依赖于光解发生的纬度与季节,还受到反应基质(冰/水)的影响.以上结果揭示了冰中和水中TC光化学转化行为的异同,这对于准确评价寒冷环境中此类新型污染物的归趋具有重要意义.
关键词:冰环境光化学水环境光化学四环素溶解性物质寒冷环境
Comparison of photodegradation for tetracycline in ice and in water: Kinetics, and effects of main water constituents
ZHANG Shuang1,2,3
 , ZHANG Peng2,3, GE Linke2,3
, ZHANG Peng2,3, GE Linke2,3
 , DONG Qianqian1,3, WANG Degao1
, DONG Qianqian1,3, WANG Degao1

1. College of Environmental Science and Engineering, Dalian Maritime University, Dalian 116026;
2. School of Environmental Science and Engineering, Shaanxi University of Science & Technology, Xi'an 710021;
3. State Environmental Protection Key Laboratory of Coastal Ecosystem, National Marine Environmental Monitoring Center, Dalian 116023
Received 10 November 2019; received in revised from 8 January 2020; accepted 8 January 2020
Abstract: Ice and snow are important environmental media, ice/snow photochemistry is therefore an emerging subject in environmental sciences. In order to reveal the differences and similarities between ice and aqueous photochemistry, photochemical behavior of organic pollutants in ice and in water requires comparative studies. In this study, we selected tetracycline (TC) as a case to compare photodegradation kinetics in ices and in waters from different water bodies under the simulated sunlight (λ>290 nm) by considering the effects of dissolved substances (humic acid (HA), Cl-, NO3- and Fe(III)). It was observed that the photodegradation of TC followed pseudo-first-order kinetics with the higher quantum yield (ΦS=4.76×10-3) in pure-water ice than in pure water (ΦS=3.85×10-3). TC underwent the fastest photolysis in seawater ice followed by freshwater ice and pure-water ice; while TC photodegraded faster in freshwater and seawater than in pure water. Further studies found that Cl-, HA, NO3- and Fe(III) accelerated the photodegradation of TC, with higher efficiencies under higher concentrations, in both ice and water phases. However, the promotion degrees for each factors are different, and HA, NO3- and Fe(III) showed stronger sensitization in ice than in water, to promote TC photodegradation. While Cl- in ice promoted less. The positive effects of these constituents on TC photolysis explained most of the stronger photodegradation potential of TC in natural ices/waters than in pure-water ice or pure water. Furthermore, extrapolation of the lab data to the real environment indicated that the photolytic half-lives of TC in mid-winter of 35°~50°N latitude were estimated to be 15.4~38.9 min and 19.0~48.0 min, respectively in ice/snow surface and in surface water. The half-lives depend not only on the latitude and season, but also on the reaction matrixes (ice/water). The differences and similarities of TC photochemical behavior in ice and in water uncovered by this study would better assess the fate and risk of such emerging pollutants in the cold environments.
Keywords: ice photochemistryaqueous photochemistrytetracyclinewater constituentscold environment
1 引言(Introduction)冰雪是一类重要的环境介质, 地球表面每年被冰雪覆盖的总面积占地球表面积的11%~23%, 中国冰雪面积占中国国土面积的1%~44%, 渤海海冰面积甚至超过1/3(孙鸿烈, 2000).较多研究表明, 冰雪中含有大量的有机污染物及光化学活性物质, 或者水体中的物质经结冰作用而封存于冰中(Na et al., 2011a; Fuoco et al., 2012; Pu?ko et al., 2015; Cipro et al., 2017; Lebedev et al., 2018).污染物在冰雪内部的物理迁移过程、常规的化学反应及微生物作用相对较弱, 而受温度影响较小的光化学作用则表现出特殊的重要性, 光化学转化已成为冰雪表层许多有机污染物重要的消减途径(康春莉等, 2008; Ram et al., 2009; Kahan et al., 2010; Ge et al., 2016a).并且由此形成了一门新的学科—冰雪环境光化学, 其主要研究冰雪中有机污染物的光化学转化动力学、影响因素、转化产物、光致毒性等关键科学问题, 以揭示寒冷环境中污染物的光化学转化和归趋.
近年来, 冰雪中有机污染物的光化学行为逐渐被广泛关注, 有关其转化动力学、影响因素、降解产物等相关研究的水平持续提升, 模型化合物的种类数量和复杂程度也在增加(Rowland et al., 2011; 葛林科等, 2015a; Hullar et al., 2018; Xue et al., 2019a).然而, 相对于水环境光化学, 冰雪环境光化学的研究深度和广度还远远不够, 并且这两种反应相中的光化学行为异同有待深入揭示.在相同光照条件下, 比较研究冰中和水中同一污染物的光化学行为, 有利于揭示不同反应相及其溶解性物质(如溶解性有机质(DOM)、Cl-、
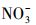
由于抗生素的大量使用和排放, 其作为新型污染物在环境水体中不断被监测到, 检出率较高的为四环素类(TCs)、磺胺类、氟喹诺酮类、大环内酯类抗生素(Ge et al., 2010; 葛林科等, 2015b).其中, TCs是畜禽养殖业中使用量最大的一类抗生素, 因在环境中检出频繁及浓度较高而被广泛关注(Cabello, 2006).我国北方环境表层水体中四环素(TC)的浓度水平如表 1所示.秦岭-淮河线是中国南方和北方的地理分界线, 在秦岭-淮河线以北, 由于冬季较其他季节河流水流慢、温度低、易结冰, 此时抗生素最稳定, 从而使冬季此类污染物的浓度水平最高, 所以北方结冰的水体中应该广泛存在大量的TCs类污染物(Luo et al., 2010; Luo et al., 2011; 葛林科等, 2015b).然而, 有关冰中该类污染物的环境光化学行为还未见报道.目前, 可以检索到水中TCs类污染物的光化学转化及光致毒性等相关研究, 发现日光照射下, TC可以发生直接光解和敏化光解, 并对Vibrio fischeri和P.phosphoreum表现为光修饰毒性(Werner et al., 2006; Jiao et al., 2008; Niu et al., 2013).
表 1(Table 1)
| 表 1 我国北方环境表层水体中四环素的浓度水平 Table 1 Concentration levels of tetracycline in environmental surface waters from north China | ||||||||||||||||||||||||||||||||||||||||||||||||
表 1 我国北方环境表层水体中四环素的浓度水平 Table 1 Concentration levels of tetracycline in environmental surface waters from north China
| ||||||||||||||||||||||||||||||||||||||||||||||||
腐殖酸(HA)广泛存在于天然水环境中, 占水溶性有机物成分(2.2~18 mg·L-1)的60%~80%, Cl-是海水的主要溶解性成分(约0.54 mol·L-1),
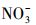
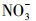
2 材料与方法(Materials and methods)2.1 实验材料四环素(TC)购于北京百灵威科技有限公司, 纯度>98%;对硝基苯甲醚(PNA, 纯度>97%)购于英国Alfa Aesar A Johnson Matthey公司; 乙腈、三氟乙酸、吡啶(Pyr)为色谱纯(美国TEDIA), 纯水由美国Millipore-Milli Q系统制备; 腐殖酸钠盐(Humic acid sodium salt, HASS)购于Aldrich公司(美国); 其他试剂NaCl、NaNO3、Fe2(SO4)3均为分析纯, 购自天津市科密欧化学试剂有限公司.淡水和海水分别取自于大连凌水西山水库和星海公园海域, 水样过0.22 μm滤膜, 以去除颗粒物和浮游生物等, -20 ℃保存.Shimadzu Class-VP离子色谱和Optima 2000 DV电感耦合等离子体-原子发射光谱分别用于测定过滤后天然水样中的阴离子和阳离子(Ge et al., 2009), 其含量参见表 2.
表 2(Table 2)
| 表 2 过0.22 μm滤膜后淡水、海水样品的性质和主要离子含量 Table 2 Properties and main ion contents of freshwater and seawater samples filtered by 0.22 μm filters | ||||||||||||||||||||||||||||||
表 2 过0.22 μm滤膜后淡水、海水样品的性质和主要离子含量 Table 2 Properties and main ion contents of freshwater and seawater samples filtered by 0.22 μm filters
| ||||||||||||||||||||||||||||||
2.2 光解实验2.2.1 冰中TC的光降解将纯水、淡水、海水配制的TC溶液(C0=5.0 μmol·L-1, V=40 mL)倒入石英试管中, 放置于暗室条件下, -20 ℃冷冻至完全结冰.采用改制的恒温(-(5±1) ℃)冷冻光解反应箱, 其中放入旋转式光化学反应仪(图 1).光源为500 W高压汞灯, 用Pyrex双层套管通循环水进行冷却, 光源截止波长λ>290 nm.对冰冻后的样品进行光照, 以PNA/Pyr为化学露光计, 并通过公式(1)计算TC的量子产率(Φs)(Dulln et al., 1982; Edhlund et al., 2006).
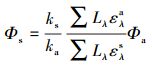 | (1) |
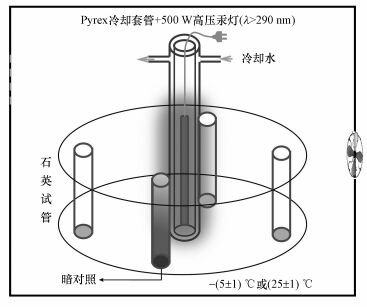 |
| 图 1 光化学实验装置示意图 Fig. 1Schematic diagram of photochemical experimental device |
式中, 下标(上标)a表示露光计; 下标(上标)s表示模型化合物; 量子产率Φa = 0.44CPyr+0.00028;k为光降解速率常数(min-1); Lλ为波长λ处的光源光强(mW·cm-2); ελ为模型化合物(上标s)或露光计(上标a)的摩尔吸光系数(L·mol-1·cm-1).
使用UV-A紫外辐照计(北京师范大学光电仪器厂)测得样品在365 nm处的光强为4.60 mW·cm-2, 在420 nm处的光强为3.26 mW·cm-2.进一步, 根据溶解性物质的环境浓度水平(Wu et al., 2000; 周成智, 2018)并结合水样检测结果(表 2), 在四环素的纯水冰溶液中分别添加NaCl、HASS、NaNO3和Fe2(SO4)3, 开展光照实验, 以研究溶解性物质Cl-、HASS、
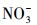
2.2.2 水中TC的光降解在相同的光照条件和反应仪(图 1)上, 开展纯水、淡水和海水中TC的光降解实验.实验在通风橱里进行, 保持通风, 并控制反应温度为(25±1) ℃.同样, 纯水中添加溶解性物质, 以考察其对TC光解动力学的影响.
2.2.3 样品分析TC浓度采用Waters超高效液相色谱仪(UPLC)进行检测, 其中, 色谱柱为BEHC18柱(2.1 mm×50 mm, 1.7 μm), 柱温为25 ℃; 三氟乙酸水溶液(pH=2.4)和纯乙腈为流动相(体积比为80 : 20), 流速为0.15 mL·min-1, 进样量为10 μL, 紫外检测波长为355 nm, 保留时间为3.0 min.
3 结果与讨论(Results and discussion)3.1 不同冰/水中四环素的表观光解在暗对照实验中, 四环素(TC)几乎没有降解, 而在模拟太阳光(λ>290 nm)照射下, 不同冰中和水中TC均出现了明显的光降解.进一步拟合并验证ln(C/C0)-t的线性关系, 即:
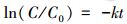 | (2) |
通过比较不同水体冰相和水相中TC光降解动力学的差异(图 2), 发现对于冰相, TC在海水冰中光解最快, 淡水冰中次之, 纯水冰中最慢.而对于水相, 光解快慢顺序与冰相不同, 为淡水中>海水中>纯水中.不同水中溶解性物质组成不同, 并且结冰过程影响其光化学活性(Grannas et al., 2014; Fede et al., 2015), 因此可以推测, 不同相TC光解快慢顺序的差异可能依赖于水中不同的溶解性物质.由表 2可知, Cl-是海水中的主要成分, 腐殖酸(HA)是淡水中主要的溶解性物质,
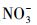
图 2(Fig. 2)
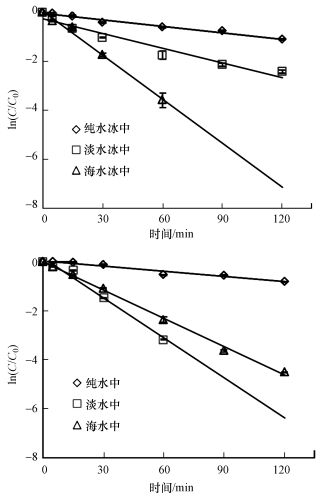 |
| 图 2 不同水体冰相(a)和水相(b)中四环素的光降解动力学拟合曲线 Fig. 2The photodegradation kinetic fitting curves of tetracycline in different ice(a) and water(b) phases |
3.2 冰/水中主要溶解性物质对四环素光解动力学的影响考察了不同浓度梯度的Cl-、HASS、
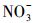
表 3(Table 3)
| 表 3 不同溶液条件下冰相与水相四环素的光降解半减期(t1/2) Table 3 Photolytic half-lives(t1/2) of tetracycline in ice/water phases containing different dissolved constituents | |||||||||||||||||||||||||||||||||||||||||||||||
表 3 不同溶液条件下冰相与水相四环素的光降解半减期(t1/2) Table 3 Photolytic half-lives(t1/2) of tetracycline in ice/water phases containing different dissolved constituents
| |||||||||||||||||||||||||||||||||||||||||||||||
图 3(Fig. 3)
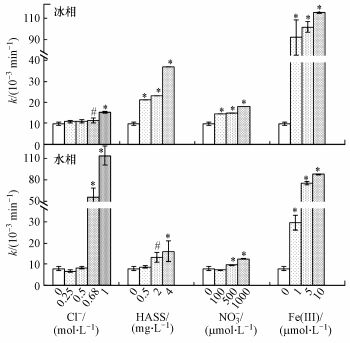 |
| 图 3 模拟日光(λ>290 nm)照射下冰/水中主要溶解性物质对对四环素光降解速率常数(k)的影响(*和#分别表示在0.05和0.1显著性水平, 与未添加溶解性物质时相比, 存在显著差异) Fig. 3Effects of main dissolved matter on the ice/aqueous photodegradation rate constant(k) of tetracycline under simulated sunlight irradiation(λ>290 nm) |
从图 3可以看出, 无论是在冰相中还是水相中, Cl-、HASS、
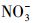

水中溶解性物质对TC光解动力学的影响, 在前人的研究中已有一些报道, 例如, Niu等(2013)和Jiao等(2008)均发现在太阳光或模拟日光照射下, 一定浓度的HA和
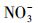
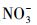
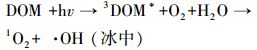 | (3) |
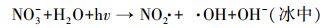 | (4) |
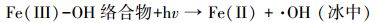 | (5) |
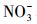
天然冰/水含有大量的阴离子、阳离子和溶解性有机质, 其中, Cl-、HA、
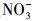
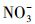
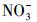
3.3 环境表层冰雪和水体中TC光降解半减期的评估在实验室模拟日光(λ>290 nm)照射下, 测得纯水冰和纯水中四环素(TC)的光解量子产率(Φs)分别为4.76×10-3和3.85×10-3.由此, 根据公式(6)~(7), 可以估算出实际环境中冰雪表面或水体表层TC的表观光解速率常数(kE)和半减期(t1/2, E).
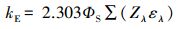 | (6) |
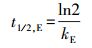 | (7) |
表 4(Table 4)
| 表 4 北纬环境中冰雪表面和水体表层四环素的表观光降解半减期(t1/2, E) Table 4 Environmental half-lives(t1/2, E) for the apparent photodegradation of tetracycline in natural surface water and ice/snow | |||||||||||||||||||||||||
表 4 北纬环境中冰雪表面和水体表层四环素的表观光降解半减期(t1/2, E) Table 4 Environmental half-lives(t1/2, E) for the apparent photodegradation of tetracycline in natural surface water and ice/snow
| |||||||||||||||||||||||||
由表 4可知, 北纬35°~50°地区冰雪表面TC的表观光解t1/2, E为15.4~38.9 min.由于冰雪环境温度低, 微生物降解作用微弱, 而光降解受温度影响较小, 所以光降解可能是中国北方冰雪表面TC等抗生素类污染物的主要消减方式.除了表观光解, TC还可能发生溶解性物质引发的敏化光解与光致ROS氧化, 并且TC是一类典型的可解离有机污染物, 可与金属离子配合(Ge et al., 2018).因此, 为全面评估结冰水体中TC等可解离抗生素的复合光化学行为, 有必要综合研究其在冰中不同解离形态的复合光化学转化动力学、产物和风险, 以及多种溶解性物质共存时的复合效应.
4 结论(Conclusions)1) 在模拟太阳光(λ>290 nm)照射下, TC发生了明显的光降解.冰相TC的光降解快慢顺序为海水冰中>淡水冰中>纯水冰中, 而水相中光解快慢顺序与冰相不同, 为淡水中>海水中>纯水中.考虑到冰/水中各种溶解性物质的相对含量及光化学活性, Cl-、腐殖酸(HA)、
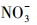
2)无论是在冰相还是水相, Cl-、HA、
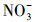
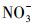
3) 在35°~50°N地区的仲冬季, 冰雪表面和水体表层TC光化学降解的t1/2, E分别为15.4~38.9 min和19.0~48.0 min.t1/2, E不仅取决于光解发生的纬度与季节, 还受到反应基质(冰/水)的影响.在仲冬同一纬度, 冰雪表面TC的光降解比在表层水体中快.
参考文献
| Blaszczak-Boxe C S, Saiz-Lopez A. 2018. Nitrate photolysis in ice and snow:A critical review of its multiphase chemistry[J]. Atmospheric Environment, 193: 224-241. DOI:10.1016/j.atmosenv.2018.09.002 |
| Cabello F C. 2006. Heavy use of prophylactic antibiotics in aquaculture:A growing problem for human and animal health and for the environment[J]. Environmental Microbiology, 8(7): 1137-1144. DOI:10.1111/j.1462-2920.2006.01054.x |
| Chen Y, Li H, Wang Z, et al. 2011. Photoproducts of tetracycline and oxytetracycline involving self-sensitized oxidation in aqueous solutions:Effects of Ca2+ and Mg2+[J]. Journal of Environmental Sciences, 23(10): 1634-1639. DOI:10.1016/S1001-0742(10)60625-0 |
| Cipro C V, Taniguchi S, Montone R C. 2017. Organic pollutants in snow and seasonal melting water from King George Island, Antarctica[J]. Water, Air, & Soil Pollution, 228(4): 149. |
| Corrochano P, Nachtigallová D, Klán P. 2017. Photooxidation of aniline derivatives can be activated by freezing their aqueous solutions[J]. Environmental Science & Technology, 51(23): 13763-13770. |
| Dulln D, Mill T. 1982. Development and evaluation of sunlight actinometers[J]. Environmental Science & Technology, 16(11): 815-820. DOI:10.1021/es00105a017 |
| Edhlund B L, Arnold W A, McNeill K. 2006. Aquatic photochemistry of nitrofuran antibiotics[J]. Environmental Science & Technology, 40(17): 5422-5427. |
| Fede A, Grannas A M. 2015. Photochemical production of singlet oxygen from dissolved organic matter in ice[J]. Environmental Science & Technology, 49(21): 12808-12815. |
| Fuoco R, Giannarelli S, Onor M, et al. 2012. A snow/firn four-century record of polycyclic aromatic hydrocarbons (PAHs) and polychlorobiphenyls (PCBs) at Talos Dome (Antarctica)[J]. Microchemical Journal, 105(11): 133-141. |
| Ge L K, Chen J W, Qiao X L, et al. 2009. Light-source-dependent effects of main water constituents on photodegradation of phenicol antibiotics:Mechanism and kinetics[J]. Environmental Science & Technology, 43(9): 3101-3107. DOI:10.1021/es8031727 |
| Ge L K, Chen J W, Wei X X, et al. 2010. Aquatic photochemistry of fluoroquinolone antibiotics:Kinetics, pathways, and multivariate effects of main water constituents[J]. Environmental Science & Technology, 44(7): 2400-2405. DOI:10.1021/es902852v |
| Ge L K, Dong Q Q, Halsall C, et al. 2018. Aqueous multivariate phototransformation kinetics of dissociated tetracycline:Implications for the photochemical fate in surface waters[J]. Environmental Science & Pollution Research, 25: 15726-15732. DOI:10.1007/s11356-018-1765-0 |
| Ge L K, Li J, Na G S, et al. 2016a. Photochemical degradation of hydroxy PAHs in ice:Implications for the polar areas[J]. Chemosphere, 155: 375-379. DOI:10.1016/j.chemosphere.2016.04.087 |
| Ge L K, Na G S, Chen C E, et al. 2016b. Aqueous photochemical degradation of hydroxylated PAHs:Kinetics, pathways, and multivariate effects of main water constituents[J]. Science of the Total Environment, 547: 166-172. DOI:10.1016/j.scitotenv.2015.12.143 |
| 葛林科, 任红蕾, 霍城, 等. 2015a. 冰中9-羟基芴的光化学降解[J]. 中国科学:化学, 45(6): 655-661. |
| 葛林科, 任红蕾, 鲁建江, 等. 2015b. 我国环境中新兴污染物抗生素及其抗性基因的分布特征[J]. 环境化学, 34(5): 875-883. |
| Grannas A M, Pagano L P, Pierce B C, et al. 2014. Role of dissolved organic matter in ice photochemistry[J]. Environmental Science & Technology, 48(18): 10725-10733. |
| Hullar T, Magadia D, Anastasio C. 2018. Photodegradation rate constants for anthracene and pyrene are similar in/on ice and in aqueous solution[J]. Environmental Science & Technology, 52(21): 12225-12234. |
| Jia J, Guan Y J, Cheng M Q, et al. 2018. Occurrence and distribution of antibiotics and antibiotic resistance genes in Ba River, China[J]. Science of the Total Environment, 642: 1136-1144. DOI:10.1016/j.scitotenv.2018.06.149 |
| Jiao S J, Zheng S R, Yin D Q, et al. 2008. Aqueous photolysis of tetracycline and toxicity of photolytic products to Luminescent bacteria[J]. Chemosphere, 73(3): 377-382. DOI:10.1016/j.chemosphere.2008.05.042 |
| Kahan T F, Donaldson D J. 2010. Benzene photolysis on ice:Implications for the fate of organic contaminants in the winter[J]. Environmental Science & Technology, 44(10): 3819-3824. DOI:10.1021/es100448h |
| 康春莉, 高红杰, 郭平, 等. 2008. 雪冰中有机污染物的研究进展[J]. 环境科学, 29(2): 468-472. |
| 郎朗, 狄静波, 董晓琪. 2018. SPE-HPLC法测定松花江哈尔滨段4种四环素类抗生素[J]. 给水排水, 54(8): 29-31. |
| Lebedev A T, Mazur D M, Polyakova O V, et al. 2018. Semi volatile organic compounds in the snow of Russian Arctic islands:Archipelago Novaya Zemlya[J]. Environmental Pollution, 239: 416-427. DOI:10.1016/j.envpol.2018.03.009 |
| Leifer A. 1988. The Kinetics of Environmental Aquatic Photochemistry:Theory and Practice[M]. Washington, DC: American Chemical Society. |
| Liu F, Liu X, Zhao S, et al. 2019. Photochemical transformations of tetracycline antibiotics influenced by natural colloidal particles:Kinetics, factor effects and mechanisms[J]. Chemosphere, 235: 867-875. DOI:10.1016/j.chemosphere.2019.06.201 |
| Luo Y, Mao D Q, Rysz M, et al. 2010. Trends in antibiotic resistance genes occurrence in the Haihe River, China[J]. Environmental Science & Technology, 44(19): 7220-7225. |
| Luo Y, Xu L Q, Rysz M, et al. 2011. Occurrence and transport of tetracycline, sulfonamide, quinolone, and macrolide antibiotics in the Haihe River Basin, China[J]. Environmental Science & Technology, 45(5): 1827-1833. |
| Malley P P, Grossman J N, Kahan T F. 2017. Effects of chromophoric dissolved organic matter on anthracene photolysis kinetics in aqueous solution and ice[J]. The Journal of Physical Chemistry A, 121(40): 7619-7626. DOI:10.1021/acs.jpca.7b05199 |
| Na G S, Fang X D, Cai Y Q, et al. 2013. Occurrence, distribution, and bioaccumulation of antibiotics in coastal environment of Dalian, China[J]. Marine Pollution Bulletin, 69(1/2): 233-237. |
| Na G S, Liu C Y, Wang Z, et al. 2011a. Distribution and characteristic of PAHs in snow of Fildes Peninsula[J]. Journal of Environmental Sciences, 23(9): 1445-1451. DOI:10.1016/S1001-0742(10)60605-5 |
| Na G S, Gu J, Ge L K, et al. 2011b. Detection of 36 antibiotics in coastal waters using high performance liquid chromatography-tandem mass spectrometry[J]. Chinese Journal of Oceanology and Limnology, 29(5): 1093-1102. DOI:10.1007/s00343-011-0225-1 |
| Niu J F, Li Y, Wang W L. 2013. Light-source-dependent role of nitrate and humic acid in tetracycline photolysis:kinetics and mechanism[J]. Chemosphere, 92(11): 1423-1429. DOI:10.1016/j.chemosphere.2013.03.049 |
| OECD.1997.Guidance Document on Direct Phototransformation of Chemicals in Water[S].Paris: OECD Environmental Health and Safety Publication |
| 彭全材, 宋金明, 李宁. 2018. 胶州湾表层海水中6类抗菌药物的分布来源与生态风险[J]. 海洋学报, 40(10): 71-83. |
| Pu?ko M, Stern G A, Macdonald R W, et al. 2015. The delivery of organic contaminants to the Arctic food web:Why sea ice matters[J]. Science of the Total Environment, 506: 444-452. DOI:10.1016/j.scitotenv.2014.11.040 |
| Ram K, Anastasio C. 2009. Photochemistry of phenanthrene, pyrene, and fluoranthene in ice and snow[J]. Atmospheric Environment, 43(14): 2252-2259. DOI:10.1016/j.atmosenv.2009.01.044 |
| Rowland G A, Bausch A R G A M. 2011. Photochemical processing of aldrin and dieldrin in frozen aqueous solutions under arctic field conditions[J]. Environmental Pollution, 159(5): 1076-1084. DOI:10.1016/j.envpol.2011.02.026 |
| 孙鸿烈. 2000. 中国资源科学百科全书[M]. 北京: 中国大百科全书出版社+石油大学出版社. |
| Weber J, Kurková R, Klánová J, et al. 2009. Photolytic degradation of methyl-parathion and fenitrothion in ice and water:Implications for cold environments[J]. Environmental Pollution, 157(12): 3308-3313. DOI:10.1016/j.envpol.2009.05.045 |
| Werner J J, Arnold W A, McNeill K. 2006. Water hardness as a photochemical parameter:Tetracycline photolysis as a function of calcium concentration, magnesium concentration, and pH[J]. Environmental Science & Technology, 40(23): 7236-7241. DOI:10.1021/es060337m |
| Wu F, Deng N S. 2000. Photochemistry of hydrolytic iron (III) species and photoinduced degradation of organic compounds.A minireview[J]. Chemosphere, 41(8): 1137-1147. DOI:10.1016/S0045-6535(00)00024-2 |
| Xue H, Zheng N, Kang C, et al. 2019a. Photochemical degradation of nitrated PAHs in snow[J]. Atmospheric Environment, 199: 260-264. DOI:10.1016/j.atmosenv.2018.11.026 |
| Xue S, Sun J, Liu Y, et al. 2019b. Effect of dissolved organic matter fractions on photodegradation of phenanthrene in ice[J]. Journal of Hazardous Materials, 361: 30-36. DOI:10.1016/j.jhazmat.2018.08.072 |
| 杨常青, 王龙星, 侯晓虹, 等. 2012. 大辽河水系河水中16种抗生素的污染水平分析[J]. 色谱, 30(8): 756-762. |
| 周成智.2018.黄河三角洲水体中有机微污染物的光解动力学模型构建[D].大连: 大连理工大学 http://cdmd.cnki.com.cn/Article/CDMD-10141-1018133404.htm |