摘要/Abstract
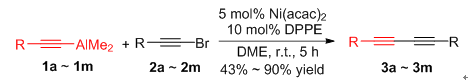
1,3-二炔类化合物是一类重要的有机合成中间体,被广泛应用于药物化学、有机合成及材料科学中.在室温条件下,以乙二醇二甲醚(DME)为溶剂,炔基溴(1 mmol)与炔基铝试剂(1.5 mmol)在Ni(acac)2(5 mol%)/DPPE(10 mol%)催化下进行偶联反应,以中等至优秀的收率得到了多种1,3-二炔烃类化合物.该反应体系对于带不同种类的功能基团的芳炔基铝试剂及炔基溴均有很好的催化活性.而且对于大位阻的α-萘炔及杂环2-噻吩炔也有较好的催化效果.该反应体系简单、催化效率高,不需要其它共催化剂.
关键词: 1,3-二炔, 金属镍, 炔基溴, 炔基铝, 偶联反应
1,3-Diyne compounds are important inter mediates in organic synthesis, which are widely used in pharmaceutical chemistry, organic synthesis and materials science. A highly efficient for the synthesis of 1,3-diyne via cross-coupling of alkynyl bromides with alkynyl aluminium reagents catalyzed by nickel has been developed. The coupling reaction of alkynyl bromides with alkynyl aluminium reagents mediated by Ni(acac)2 (5 mol%)/1,2-bis(diphenylphosphino)ethane nickel(Ⅱ) chloride (DPPE) (10 mol%)in 1,2-dimethoxyethane afforded the corresponding coupling products 1,3-diyne in good to excellent yields(up to 90%) at room temperature for 5 h. The coupling reaction of alkynyl aluminum with different substituents and alkynyl bromine with various substituents can afforded the coupling products in good yields. Importantly, the α-ethynylnaphthalene and 2-ethynylthiophene were also suitable for the reaction. This process is simple and easily performed, which provides an efficient method for the synthesis of 1,3-diynes derivatives.
Key words: 1,3-diyne, nickel, alkynyl bromide, alkynyl aluminium, cross-coupling
PDF全文下载地址:
点我下载PDF
